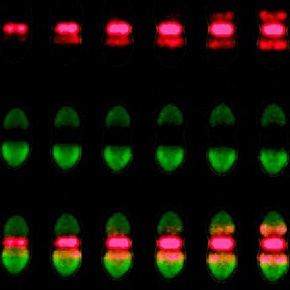
Understanding the precise mechanisms of bacterial division in streptococci is more than a matter of academic curiosity; it is a critical frontier in the fight against some of the most dangerous neonatal infections. When bacteria divide, they must accurately position the site of separation to ensure the survival and proliferation of the colony. Recent research highlighted by the Centre national de la recherche scientifique (CNRS) indicates that a transient signal is responsible for positioning this division site in streptococci, a discovery that provides a deeper appear into the cellular biology of these pathogens.
For clinicians and public health experts, the ability of these bacteria to multiply and invade host tissues is the primary driver of disease severity. In the case of Group B Streptococcus (GBS), this process can lead to devastating outcomes for newborns, including sepsis and meningitis. By uncovering how these organisms manage their physical division, scientists move closer to identifying vulnerabilities that could be targeted to halt bacterial growth.
The urgency of this research is underscored by the emergence of hypervirulent strains. As these bacteria evolve, they develop more sophisticated ways to breach the blood-brain barrier and evade the immune system. Understanding the intersection between cellular division and virulence factors is essential for developing the next generation of antimicrobial therapies.
The Threat of Group B Streptococcus in Neonatal Health
Group B Streptococcus remains a significant cause of morbidity and mortality in newborns. These infections often emerge during childbirth or shortly after, leading to systemic infections that can rapidly progress. One of the most severe manifestations is neonatal meningitis, where the bacteria penetrate the central nervous system, causing inflammation and potential long-term neurological damage.
The emergence of these infections is a complex process involving the bacteria’s ability to colonize the maternal tract and subsequently infect the neonate. According to the Institut Pasteur, explaining the emergence of these infections is key to improving prevention and treatment strategies for newborns globally.
Hypervirulence and the CC17 Strain
Not all strains of Streptococcus are created equal. Researchers have identified specific clonal complexes, such as CC17, that exhibit increased virulence. The CC17 group B Streptococcus is particularly noted for its ability to cause neonatal meningitis. This strain does not rely on chance; it actively exploits host mechanisms to facilitate its invasion.
Specifically, CC17 GBS exploits integrins—proteins that typically help cells adhere to one another or to the extracellular matrix—to develop neonatal meningitis. As detailed in research published via JCI.org, the exploitation of these integrins is a pivotal step in the bacteria’s ability to breach protective barriers and enter the bloodstream and brain.
The Role of Srr2 and Molecular Adhesion
Beyond the exploitation of integrins, hypervirulent strains like ST-17 employ specialized proteins to anchor themselves and survive within the host. One such protein is Srr2, a multifaceted adhesin expressed by ST-17 hypervirulent Group B Streptococcus.

Srr2 plays a dual role in the bacteria’s survival strategy by binding to both fibrinogen and plasminogen. This ability allows the bacteria to manipulate the host’s clotting and fibrinolysis systems, which can aid in the masking of the bacteria from the immune system and facilitate their spread through tissues. This mechanism is documented in studies hosted by the Wiley Online Library.
Connecting Cellular Division to Pathogenesis
While the study of adhesins like Srr2 and the exploitation of integrins explains how bacteria invade, the research into bacterial division in streptococci explains how they persist. The discovery by the CNRS that a transient signal positions the division site suggests a highly regulated process of growth.
If the division site is incorrectly positioned, the bacteria may fail to divide or produce non-viable offspring. By identifying the signal that ensures precise division, researchers can potentially locate ways to disrupt this process. If the “transient signal” can be blocked or mimicked, the bacteria’s ability to multiply within the neonate’s body could be severely impaired, regardless of how effectively they apply integrins or Srr2 to invade.
Key Takeaways for Public Health
- Division Precision: Streptococci rely on a transient signal to accurately position their division site, a process essential for their growth.
- Strain Specificity: The CC17 strain is particularly dangerous in neonatal cases, utilizing integrins to facilitate the development of meningitis.
- Adhesin Function: The Srr2 protein in ST-17 strains binds fibrinogen and plasminogen, contributing to the bacteria’s hypervirulence.
- Therapeutic Potential: Understanding both the invasion mechanisms (integrins/Srr2) and the growth mechanisms (division signals) provides multiple targets for future medical innovation.
The ongoing study of these pathogens highlights the necessity of a multi-pronged approach to infectious disease. We cannot rely solely on traditional antibiotics, especially as resistance grows. Instead, targeting the fundamental biological processes of the bacteria—such as their ability to divide or their method of adhering to host cells—offers a promising path forward.
Further official updates on Group B Streptococcus prevention and neonatal care guidelines are typically provided by national health agencies and the World Health Organization. Monitoring the evolution of strains like CC17 remains a priority for global health surveillance.
Do you have questions about neonatal health or the latest in infectious disease research? Share your thoughts in the comments below or share this article with your professional network.