The landscape of metastatic breast cancer treatment has been significantly altered by the introduction of CDK4/6 inhibitors, a class of targeted therapies designed to block the proteins that drive cell division. For patients with hormone receptor-positive (HR+), human epidermal growth factor receptor 2-negative (HER2-) breast cancer, these medications offer a way to delay the need for chemotherapy and improve progression-free survival.
Yet, the clinical success of optimizing CDK4/6 inhibitor use depends on more than just the prescription; it requires a precise approach to dosing, vigilant safety monitoring, and a multidisciplinary effort to manage side effects. As these drugs are often administered in combination with endocrine therapies, the complexity of the regimen increases the necessity for expert oversight to ensure patient adherence and quality of life.
As a physician and health journalist, I have observed that the role of the healthcare team—particularly the pharmacist—has shifted from simple dispensing to active clinical management. By bridging the gap between the oncologist’s prescription and the patient’s daily experience, pharmacists are now essential in identifying adverse reactions early and adjusting dosages to maintain the therapeutic window without compromising patient safety.
Understanding the Role of CDK4/6 Inhibitors in Breast Cancer
Cyclin-dependent kinase 4 and 6 (CDK4/6) are enzymes that act as “gatekeepers” for the cell cycle. In many breast cancers, these enzymes are overactive, allowing cancer cells to divide and multiply rapidly. Inhibitors specifically target these proteins to halt the cell cycle in the G1 phase, effectively stopping the growth of the tumor. These agents are typically used in combination with aromatase inhibitors or fulvestrant to create a dual blockade of the pathways that fuel cancer growth.
The clinical application of these inhibitors is evolving. For example, recent research is exploring the combination of these drugs with other targeted therapies to overcome resistance. One such development is the INAVO120 Phase III Study, which investigates the use of Invavolisib in combination with palbociclib and fulvestrant specifically for patients with PIK3CA-mutant, HR+/HER2- locally advanced or metastatic breast cancer.
Dosing Strategies and Safety Considerations
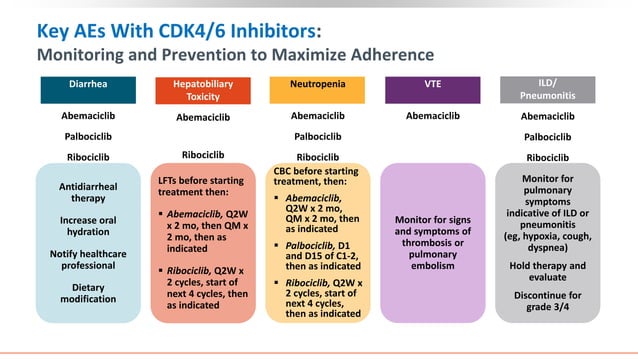
Optimizing the delivery of CDK4/6 inhibitors requires a careful balance between efficacy and toxicity. Dosing is not “one size fits all”; it often requires adjustments based on the patient’s hematologic response and the emergence of side effects. The most common concern with this class of medication is neutropenia—a significant drop in white blood cell counts—which can leave patients vulnerable to infections.
Safety management involves regular blood monitoring to track absolute neutrophil counts (ANC). If neutropenia becomes severe, clinicians may implement “dose interruptions” or “dose reductions.” The goal is to allow the bone marrow to recover without completely stopping the treatment, thereby maintaining the pressure on the cancer cells although protecting the patient’s immune system.
Beyond neutropenia, patients may experience fatigue, gastrointestinal issues, and other systemic effects. The management of these symptoms is critical because poor symptom control often leads to premature discontinuation of the therapy, which can negatively impact the overall prognosis of the disease.
The Critical Impact of Pharmacist Management
Pharmacists have become indispensable in the administration of complex oncology regimens. Their expertise allows for a more granular level of care, focusing on the practicalities of medication adherence and the mitigation of drug-drug interactions. Because CDK4/6 inhibitors are often metabolized by specific liver enzymes, certain over-the-counter medications or herbal supplements can interfere with how the drug is processed by the body.
Pharmacist-led interventions typically include:
- Medication Reconciliation: Reviewing all current medications to prevent dangerous interactions that could either increase toxicity or decrease the drug’s effectiveness.
- Patient Education: Ensuring patients understand the importance of the dosing schedule and recognize the “red flag” symptoms of neutropenia, such as fever or chills.
- Adherence Support: Helping patients navigate the logistics of their treatment cycles, which often involve periods of medication followed by short breaks.
By providing this layer of support, pharmacists help reduce the burden on oncologists and empower patients to manage their treatment at home more safely. This collaborative model ensures that the focus remains on optimizing the therapeutic outcome while minimizing the impact on the patient’s daily life.
Key Takeaways for Caregivers and Patients
| Focus Area | Key Action | Why It Matters |
|---|---|---|
| Monitoring | Regular blood tests (CBC) | Detects neutropenia early to prevent infection. |
| Dosing | Strict adherence to schedules | Prevents cancer cells from recovering during “off” periods. |
| Coordination | Consulting with pharmacists | Identifies potential drug interactions and side effect triggers. |
| Safety | Immediate reporting of fever | Neutropenic fever is a medical emergency. |
As research continues to evolve, the focus remains on refining these combinations to target specific genetic mutations, such as those found in the PIK3CA pathway. The integration of new agents into existing CDK4/6 protocols represents the next frontier in personalized breast cancer care.
For those seeking the latest clinical updates, monitoring the results of ongoing Phase III trials and official regulatory announcements regarding new combination approvals is the best way to stay informed on evolving standards of care.
Do you have questions about managing targeted therapies or the role of multidisciplinary care in oncology? We encourage you to share your thoughts and experiences in the comments below.