For decades, the medical community has viewed multiple sclerosis (MS) primarily as a disease of the “insulation.” The prevailing narrative focused on the immune system attacking myelin—the fatty sheath protecting nerve fibers—leading to the characteristic flare-ups of muscle weakness and tingling that define the relapsing-remitting phase of the condition.
However, for many patients, the disease eventually enters a more challenging stage: progressive MS. In this phase, the clinical picture shifts. The unpredictable relapses often give way to a steady, relentless decline in cognitive function, memory and reasoning. This transition is driven not just by the loss of myelin, but by the actual death of neurons—the brain’s primary signaling cells. For years, the central question for neurologists has been why certain areas of the brain succumb to this degeneration while others remain resilient.
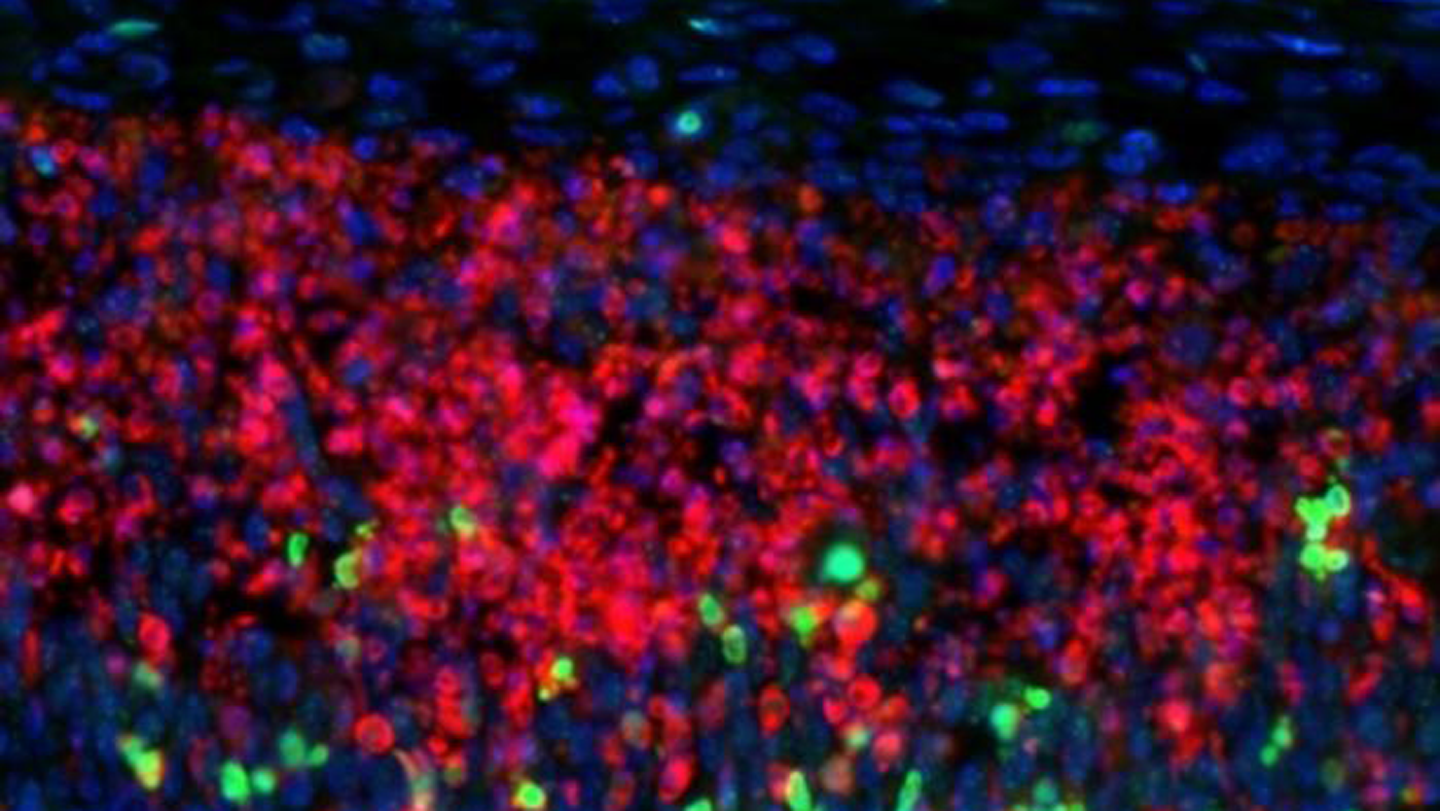
Recent research published in Nature has provided a critical answer, identifying a specific population of brain cells that are uniquely brain cells vulnerable to multiple sclerosis. These cells, known as CUX2 neurons, are essential for the complex cognitive processes that define human intelligence, but they possess a biological “blind spot” that makes them susceptible to the inflammatory environment of MS.
This discovery marks a pivotal shift in our understanding of neurodegeneration. By pinpointing the exact cellular vulnerability and the mechanism of their failure—a breakdown in DNA repair—scientists are now moving closer to developing neuroprotective therapies that could potentially halt the cognitive decline associated with progressive MS.
The Role of CUX2 Neurons in the Human Cortex
To understand why the loss of CUX2 neurons is so devastating, one must first understand where they live and what they do. These neurons are primarily located in the human cortex, the brain’s wrinkled outermost layer. Specifically, they populate the upper layers of the cortex, which are responsible for high-level integration, computation, and cognition.

Unlike the deep-brain structures that handle basic survival functions, the CUX2-positive neurons are heavily involved in the “executive” functions of the brain. When these cells are healthy, they allow us to reason, plan, and process complex information. When they begin to die, the brain physically shrinks—a process known as cortical atrophy—which manifests clinically as a decline in mental acuity and memory loss.
Research indicates that while many types of neurons are present in the brain, CUX2 neurons are disproportionately affected during the progressive phase of MS. This selectivity suggests that the disease does not attack the brain randomly; rather, it exploits a specific biological weakness inherent to these high-functioning cells.
The DNA Repair Gap: Why These Cells Die
The vulnerability of CUX2 neurons is not due to a lack of defense, but rather a failure of “maintenance” under pressure. Every cell in the human body possesses a cellular repair kit—a set of enzymes and proteins designed to fix DNA damage. Here’s a vital process because DNA is constantly being attacked by oxidative stress and inflammation.
In a healthy brain, the rate of DNA damage is balanced by the rate of repair. However, in patients with multiple sclerosis, the brain is subjected to chronic neuroinflammation. This inflammatory environment generates a constant barrage of toxins and free radicals that cause double-strand breaks in the DNA of neurons.
The breakthrough findings reveal that in CUX2 neurons, the damage caused by inflammation simply outpaces the cells’ ability to self-repair. While other neurons may be able to keep up with the repairs or activate protective pathways, CUX2 neurons reach a tipping point. Once the DNA damage exceeds the capacity of the repair mechanism, the cell triggers a programmed death sequence, known as apoptosis.
This “repair gap” explains why the cognitive decline in progressive MS is so persistent. Even if the initial inflammatory flare-up is controlled, the accumulated DNA damage in these specific neurons can lead to a delayed but inevitable cell death, contributing to the gradual shrinking of the brain.
Shifting the Paradigm: From Myelin to Neuroprotection
For the majority of MS history, treatment has focused on immunomodulation—using drugs to stop the immune system from attacking the myelin sheath. While these therapies are highly effective at reducing relapses in relapsing-remitting MS, they have historically struggled to stop the progression of the disease once it reaches the neurodegenerative phase.
The reason for this struggle is now clearer: stopping the immune attack (the cause of the damage) is not the same as repairing the neurons or preventing the cells from dying due to accumulated DNA stress. The focus must shift from “stopping the attack” to “protecting the cell.”
By identifying the DNA repair failure in CUX2 neurons, researchers have opened a new door for medical innovation. The goal is no longer just to keep the immune system at bay, but to enhance the cellular repair kits within the neurons themselves. If scientists can develop drugs that boost the efficiency of DNA repair or shield CUX2 neurons from inflammatory stress, they may be able to preserve cognitive function even in the presence of the disease.
What This Means for Patients and Families
For the estimated hundreds of thousands of people living with MS, particularly those in the progressive stages, this research offers a tangible sense of hope. The feeling of “cognitive fog” or the loss of reasoning abilities has long been one of the most distressing aspects of the disease, often feeling untreatable because the cells were already gone.
The identification of the CUX2 vulnerability transforms the cognitive decline from an inevitable consequence of the disease into a targetable biological process. While we are not yet at the stage of a “cure” for progressive MS, we have moved from not knowing why the brain shrinks to knowing exactly which cells are dying and how it is happening.
From a clinical perspective, this may eventually lead to better diagnostic tools. If we can identify the markers of DNA repair failure in the brain early on, physicians might be able to predict which patients are at higher risk for rapid cognitive decline and intervene with neuroprotective strategies before significant atrophy occurs.
The Road Ahead: From Lab to Clinic
The transition from a laboratory discovery in human brain cells and mouse models to a bedside treatment is a rigorous process. The next steps for the scientific community involve several critical phases:
- Drug Screening: Testing existing compounds that enhance DNA repair to see if they can protect CUX2 neurons from inflammatory damage.
- Biomarker Development: Finding a way to measure DNA damage or repair efficiency in living patients, perhaps through advanced imaging or cerebrospinal fluid analysis.
- Clinical Trials: Moving toward human trials to determine if neuroprotective agents can actually slow the rate of brain atrophy in progressive MS patients.
The discovery of the vulnerability of CUX2 neurons serves as a reminder that the brain is not a monolith. Different cells have different strengths and weaknesses, and the key to treating complex neurological diseases lies in understanding these cellular nuances.
The next confirmed milestone in this research trajectory will be the publication of results from early-stage pharmacological screens aimed at boosting DNA repair mechanisms in cortical neurons. As these studies progress, the medical community expects to see a new class of “neuroprotective” therapies enter the clinical pipeline, specifically designed to safeguard the cells that make us human.
Do you or a loved one manage the challenges of progressive MS? We invite you to share your experiences or questions in the comments below to help foster a community of support and shared knowledge.