A new clinical trial conducted in Nigeria has identified a promising alternative for treating Lassa fever, a viral hemorrhagic fever endemic to West Africa. The results of an open-label, randomized controlled phase 2 trial indicate that the antiviral medication favipiravir is both safe and well tolerated in patients with mild-to-moderate cases of the disease.
For years, treatment options for Lassa fever have been limited, with ribavirin serving as a primary intervention. However, the search for alternatives that can provide reliable exposure and better tolerability has remained a priority for public health officials and infectious disease specialists. This latest research provides critical data on how favipiravir behaves in the human body during an acute Lassa fever infection.
The study, which focused on patients hospitalized at two reference hospitals in Nigeria, met its primary endpoints regarding safety and pharmacokinetic parameters. Most notably, all participants in the trial survived and were successfully discharged from their isolation wards, providing a hopeful signal for the further optimization of favipiravir as a treatment alternative.
Trial Design and Patient Demographics
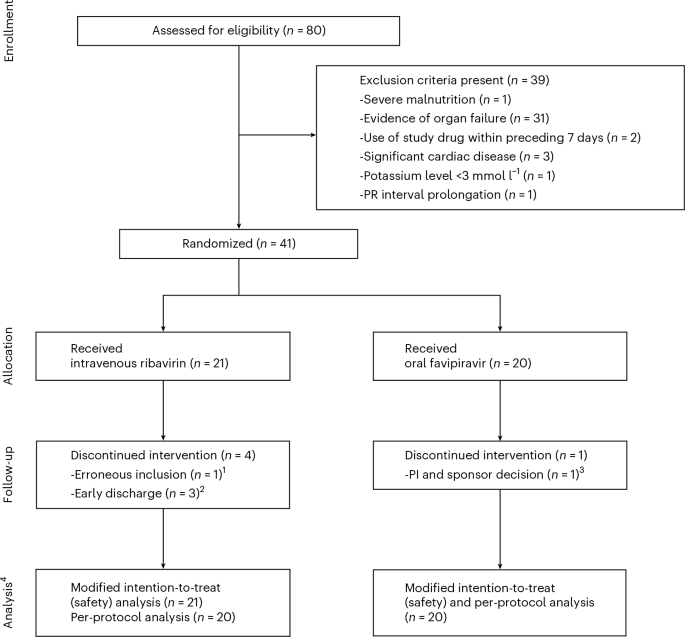
The phase 2 trial was designed as an open-label, randomized controlled study to evaluate the efficacy and safety of favipiravir compared to the traditional use of ribavirin. The researchers recruited hospitalized adult patients who had RT-PCR-confirmed mild-to-moderate Lassa fever. In total, 41 patients were randomized into two groups: 21 received ribavirin and 20 received favipiravir.

Of the initial cohort, 36 patients completed the full 10-day follow-up period. The demographic profile of the participants reflected a broad range of adults, with a median age of 37 years. Gender distribution was nearly equal, with 19 participants (46.3%) being female.
Safety and Tolerability Findings
One of the most critical aspects of any antiviral trial is the assessment of treatment-emergent adverse events (TEAEs). In this study, researchers identified a total of 30 drug-related TEAEs. These events were distributed evenly between the two treatment arms, with 16 events (53.5%) occurring in the favipiravir group.

Crucially, none of the adverse events associated with favipiravir were classified as severe or serious. The study also highlighted distinct differences in the types of side effects experienced by the two groups. While anemia was the most frequently observed adverse event among patients receiving ribavirin, vomiting was the most common side effect reported in the favipiravir arm.
Pharmacokinetic Analysis of Favipiravir
To understand how the drug is absorbed and processed by the body, the researchers utilized a one-compartment model for pharmacokinetic (PK) analysis. This analysis is essential for determining the correct dosage to ensure the drug reaches a therapeutic concentration in the bloodstream without becoming toxic.
The PK analysis of favipiravir demonstrated reliable exposure in the patients. Key findings included a maximum plasma concentration (Cmax) of 50.9 mg/L (with an interquartile range of 42.1 to 75.1 mg/L) in a steady state. The drug’s half-life—the time it takes for the concentration of the drug in the plasma to reduce by half—was measured at 10.9 hours (IQR 8.2 to 17.1 h).
the area under the curve (AUC) from 0 to 240 hours was recorded at 9,275 mg L−1 h−1 (IQR 7,139.4 to 15,794.8 mg L−1 h−1). These metrics suggest that favipiravir maintains a consistent presence in the system, which is vital for combating the replication of the Lassa virus.
Implications for Lassa Fever Management
Lassa fever remains a significant public health challenge in West Africa due to high case fatality rates among hospitalized patients. The evidence that favipiravir is safe and well tolerated in mild-to-moderate cases opens the door for its potential integration into broader treatment protocols.

By providing a viable alternative to ribavirin, healthcare providers may have more flexibility in treating patients, particularly those who may not tolerate ribavirin well. The fact that all trial participants survived and were discharged underscores the potential of this antiviral approach, though further research is needed to determine its efficacy in severe cases of the disease.
Key Takeaways from the Trial
- Safety Profile: Favipiravir was found to be safe and well tolerated, with no severe or serious drug-related adverse events reported.
- Patient Outcomes: 100% of the trial participants survived and were successfully discharged from isolation.
- Side Effect Comparison: Vomiting was the most common side effect for favipiravir, whereas anemia was most prevalent in the ribavirin group.
- Reliable Exposure: Pharmacokinetic data showed a steady-state maximum plasma concentration of 50.9 mg/L and a half-life of 10.9 hours.
- Clinical Scope: The trial focused on adult patients with RT-PCR-confirmed mild-to-moderate Lassa fever in Nigeria.
The next steps for this research involve the further optimization of favipiravir to refine dosage and administration, ensuring the best possible outcomes for patients battling this viral hemorrhagic fever. While these phase 2 results are encouraging, larger-scale trials will be necessary to fully establish favipiravir as a standard of care.
We welcome your thoughts on these developments in antiviral research. Please share this article with your colleagues in the public health community or leave a comment below to join the discussion.