The landscape of treatment for newly diagnosed multiple myeloma is evolving rapidly, with clinicians increasingly looking toward “quadruplet” therapies to deepen patient responses. A significant step forward has arrived with the results of the EMN24 IsKia trial, which evaluated the efficacy of combining isatuximab, carfilzomib, lenalidomide and dexamethasone in newly diagnosed multiple myeloma patients who are eligible for autologous stem cell transplantation.
For patients facing this complex plasma cell malignancy, the goal of treatment is not just the reduction of cancer cells, but the achievement of a state where the disease is undetectable by the most sensitive tests available. The IsKia trial specifically targeted this goal, comparing a four-drug regimen against a standard three-drug approach to witness if the addition of a CD38-targeting monoclonal antibody could significantly improve outcomes.
The findings, published recently in Nature Medicine, indicate that the quadruplet approach—referred to as Isa-KRd—leads to significantly higher rates of measurable residual disease (MRD) negativity compared to the KRd triplet. This suggests that the combined force of these four agents can clear the bone marrow of myeloma cells more effectively, potentially setting the stage for more durable remissions in transplant-eligible patients.
The Architecture of the EMN24 IsKia Trial
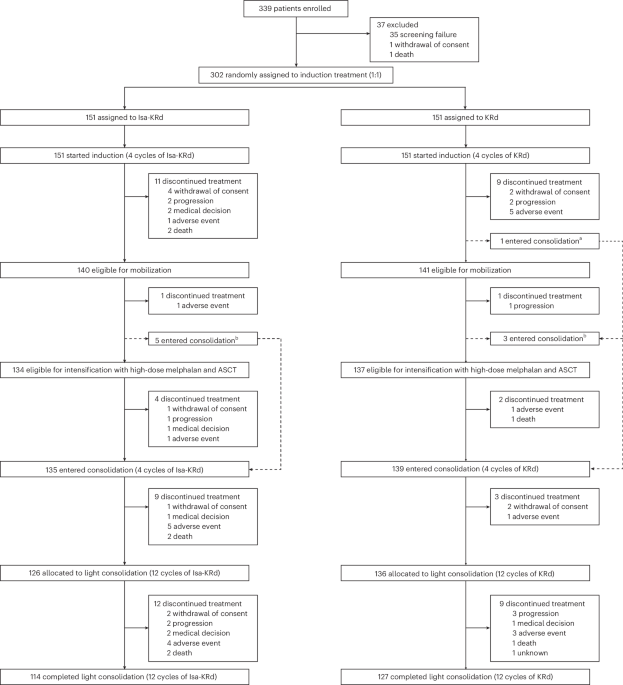
The EMN24 IsKia trial was designed as a phase III randomized study to provide a rigorous comparison between two treatment pathways. The study enrolled 302 transplant-eligible (TE) patients with newly diagnosed multiple myeloma (NDMM) who were aged 70 years or younger Nature Medicine.
Patients were randomized in a 1:1 ratio into two distinct treatment arms:
- Arm A (Isa-KRd): A quadruplet therapy consisting of isatuximab, carfilzomib, lenalidomide, and dexamethasone.
- Arm B (KRd): A triplet therapy consisting of carfilzomib, lenalidomide, and dexamethasone.
The treatment sequence was comprehensive, designed to attack the cancer at multiple stages of the patient’s journey. Both groups underwent four cycles of induction therapy, followed by an autologous stem cell transplantation (ASCT). Following the transplant, patients received four cycles of post-ASCT consolidation, and finally, 12 cycles of “light consolidation” using their respective assigned regimens European Myeloma Network.
Defining Success: The Role of MRD Negativity
To understand the significance of the IsKia trial, it is essential to understand “measurable residual disease” (MRD). In multiple myeloma, a patient may appear to be in complete remission through standard testing, but highly sensitive tools can still detect a tiny fraction of remaining cancer cells. MRD negativity means that these remaining cells are undetectable, which is often viewed as a marker for a deeper and more durable response.

The IsKia trial utilized next-generation sequencing (NGS) to measure this, focusing on two levels of sensitivity: $10^{-5}$ (one cancer cell in 100,000) and the even more stringent $10^{-6}$ (one cancer cell in one million). The primary endpoint of the study was the rate of MRD negativity at the $10^{-5}$ sensitivity level after the consolidation phase Nature Medicine.
Post-Consolidation Results
The data revealed a clear advantage for the Isa-KRd quadruplet therapy. After the consolidation phase, MRD negativity rates were significantly higher in the Isa-KRd arm compared to the KRd arm:
- At $10^{-5}$ sensitivity: 77% for Isa-KRd versus 67% for KRd (odds ratio 1.67, P = 0.049).
- At $10^{-6}$ sensitivity: 68% for Isa-KRd versus 48% for KRd (odds ratio 2.36, P = 0.0004).
These figures demonstrate that the addition of isatuximab not only increased the likelihood of reaching MRD negativity but also increased the likelihood of achieving a “deep” response (the $10^{-6}$ level), which is a critical clinical milestone Nature Medicine.
Rapid and Durable Responses
One of the most striking aspects of the IsKia trial was how quickly the quadruplet therapy worked. The study found that deep MRD responses were achieved more rapidly during the induction phase—the period before the stem cell transplant. At the $10^{-5}$ sensitivity level, 46% of Isa-KRd patients achieved MRD negativity post-induction, compared to only 27% of KRd patients (P = 0.0007). At the $10^{-6}$ level, the difference was even more pronounced: 28% for Isa-KRd versus 14% for KRd (P = 0.0029) Nature Medicine.
Beyond the speed of the response, the durability of these results is a key indicator of long-term success. The trial tracked “sustained” MRD negativity, finding that 52% of patients in the Isa-KRd group maintained $10^{-6}$ MRD negativity for one year, compared to 38% in the KRd group (odds ratio 1.82, P = 0.012) Nature Medicine.
Safety Profile and Clinical Trade-offs
When introducing a fourth drug into a chemotherapy regimen, clinicians must carefully weigh the increased efficacy against potential increases in toxicity. But, the EMN24 IsKia trial indicates that the safety profile of the quadruplet therapy remained manageable.
According to the study results, Grade 3–4 non-hematologic adverse events (AEs) were similar between the Isa-KRd and KRd arms. There were no significant differences between the two groups regarding treatment discontinuations or deaths resulting from adverse events Nature Medicine.
While the MRD data is compelling, the data for progression-free survival (PFS)—the length of time during and after treatment that a patient lives with the disease but it does not get worse—are currently immature. So that while more patients are reaching an undetectable disease state, the long-term impact on overall survival and disease progression is still being monitored.
Key Trial Comparison Summary
| Metric | Isa-KRd (Quadruplet) | KRd (Triplet) | P-Value |
|---|---|---|---|
| Post-Consolidation MRD ($10^{-5}$) | 77% | 67% | 0.049 |
| Post-Consolidation MRD ($10^{-6}$) | 68% | 48% | 0.0004 |
| Post-Induction MRD ($10^{-5}$) | 46% | 27% | 0.0007 |
| Post-Induction MRD ($10^{-6}$) | 28% | 14% | 0.0029 |
| 1-Year Sustained MRD ($10^{-6}$) | 52% | 38% | 0.012 |
What This Means for the Future of Myeloma Care
The results of the IsKia trial contribute to a growing body of evidence supporting the use of quadruplet therapies in transplant-eligible patients. By combining a proteasome inhibitor (carfilzomib), an immunomodulatory drug (lenalidomide), a steroid (dexamethasone), and a CD38 monoclonal antibody (isatuximab), physicians can more aggressively target the myeloma cells from multiple biological angles.
For patients, this may mean a higher probability of achieving “deep” responses that were previously less common with triplet therapies. The fact that these responses are achieved more quickly during induction is particularly noteworthy, as it may influence how patients are managed leading up to their stem cell transplant.
As the medical community awaits the mature progression-free survival data, the IsKia trial provides a strong signal that the Isa-KRd regimen can significantly improve NGS-MRD negativity while maintaining a safety profile comparable to the standard-of-care triplet Nature Medicine.
Detailed information regarding this study can be found via its ClinicalTrials.gov registration: NCT04483739.
The next major milestone for this research will be the release of mature progression-free survival (PFS) data, which will determine if the higher rates of MRD negativity translate directly into longer periods of disease control for patients.
Do you have questions about the latest advancements in multiple myeloma treatment? Share your thoughts in the comments or share this article with others who may find this clinical update helpful.