Semaglutide Shows Promise in Reducing Cardiovascular Events in Patients with Liver Fibrosis Risk
Berlin, Germany – A new analysis of data from the SELECT trial indicates that the medication semaglutide, already recognized for its role in weight management and cardiovascular health, may offer significant benefits to individuals at higher risk of liver fibrosis. The findings, published online today, April 2, 2026, reveal a reduction in major adverse cardiovascular events (MACE) among patients receiving semaglutide compared to those receiving a placebo, with the effect appearing more pronounced in those exhibiting signs of liver disease. This research adds another layer to the growing understanding of semaglutide’s potential beyond weight loss, potentially offering a new avenue for managing cardiovascular risk in a vulnerable population.
The SELECT trial, a randomized, placebo-controlled study, originally demonstrated a 20% reduction in MACE – encompassing cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke – in patients with established atherosclerotic cardiovascular disease and obesity, but without diabetes. This latest analysis focused on a subgroup of participants at elevated risk for substantial liver fibrosis, assessed using the Fibrosis-4 (F4) index, a non-invasive marker of liver scarring. The implications of these findings are substantial, given the increasing prevalence of both obesity and non-alcoholic fatty liver disease (NAFLD), now more accurately termed metabolic dysfunction-associated steatotic liver disease (MASLD), and their interconnected impact on cardiovascular health.
Understanding the Link Between Liver Health and Cardiovascular Disease
The relationship between liver disease and heart problems is increasingly recognized by medical professionals. Obesity is a major risk factor for the development of MASLD, which involves the accumulation of fat in the liver. This can progress to inflammation and fibrosis, ultimately leading to cirrhosis and liver failure. However, the consequences of liver damage extend far beyond the liver itself. As experts at Archynetys explain, liver scarring actively worsens cardiovascular risk, creating a complex interplay of metabolic dysfunction and hepatic pathology. The liver plays a crucial role in regulating cholesterol and other metabolic processes, and its dysfunction can contribute to the development of atherosclerosis – the buildup of plaque in the arteries – and increase the risk of heart attack and stroke.
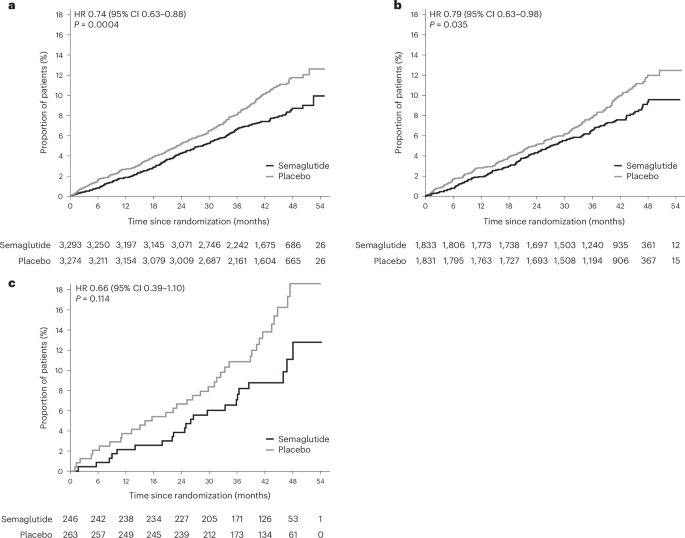
The F4 index, used in the SELECT trial analysis, provides a relatively simple and cost-effective way to identify individuals at higher risk of significant liver fibrosis. Scores of 1.3 or greater are generally considered indicative of increased risk, with higher scores correlating with more advanced fibrosis. The SELECT trial analysis examined several F4 score thresholds to assess the impact of semaglutide across different levels of risk.
Key Findings from the SELECT Trial Analysis
The prespecified analysis revealed a compelling trend: semaglutide’s protective effect against MACE appeared to be stronger in patients with higher F4 scores. Specifically, the study found a 26% reduction in MACE among those with baseline F4 scores of 1.3 or greater (hazard ratio 0.74, 95% confidence interval 0.63–0.88, P=0.0004). This benefit was also observed, though to a lesser extent, in individuals with moderately elevated F4 scores (21% reduction, hazard ratio 0.79, 95% confidence interval 0.63–0.98, P=0.035). While a reduction was seen in those with very high F4 scores (>2.67), it did not reach statistical significance (34% reduction, hazard ratio 0.66, 95% confidence interval 0.39–1.10, P=0.11).
Beyond the reduction in cardiovascular events, the analysis also demonstrated a significant impact on liver health itself. Semaglutide led to a 28% greater decrease in the fatty liver index compared to placebo (hazard ratio 0.72, 95% confidence interval 0.71–0.73, P<0.0001). The fatty liver index is another tool used to assess the risk of MASLD. This suggests that semaglutide may not only protect against the cardiovascular consequences of liver disease but also directly improve liver health.
Semaglutide and NASH-Related Cirrhosis: Further Research
The potential of semaglutide extends to even more advanced stages of liver disease. A separate study, published in The Lancet Gastroenterology & Hepatology, assessed the efficacy and safety of a higher dose of semaglutide (2.4 mg once weekly) in patients with NASH-related compensated cirrhosis (fibrosis stage 4). This phase 2 trial is the first of its kind to investigate this specific application of semaglutide, offering a promising avenue for treatment in a population with limited therapeutic options.
Implications for Clinical Practice and Future Research
These findings have significant implications for clinical practice. The data suggest that semaglutide may be particularly beneficial for patients with obesity, cardiovascular disease, and signs of liver fibrosis. However, it’s significant to note that semaglutide is not a one-size-fits-all solution. Careful patient selection and monitoring are crucial, as with any medication. Further research is needed to fully understand the long-term effects of semaglutide on liver health and cardiovascular outcomes, and to identify the optimal dosage and duration of treatment.
The SELECT trial analysis and the ongoing research into NASH-related cirrhosis highlight the evolving understanding of semaglutide’s therapeutic potential. What began as a medication primarily focused on weight loss is now emerging as a promising tool for managing a range of metabolic and cardiovascular conditions, particularly in individuals with underlying liver disease. This underscores the importance of considering the interconnectedness of these conditions and adopting a holistic approach to patient care.
Key Takeaways
- Semaglutide reduces major adverse cardiovascular events (MACE) in patients with atherosclerotic cardiovascular disease and obesity.
- The benefit of semaglutide appears to be greater in patients at higher risk of liver fibrosis, as indicated by the Fibrosis-4 index.
- Semaglutide is associated with a significant reduction in the fatty liver index, suggesting a direct improvement in liver health.
- Ongoing research is exploring the use of higher doses of semaglutide in patients with NASH-related cirrhosis.
Researchers will continue to analyze data from the SELECT trial and other studies to refine our understanding of semaglutide’s effects. The next steps include longer-term follow-up studies to assess the durability of the observed benefits and investigations into the mechanisms by which semaglutide exerts its protective effects on both the heart and the liver. The clinical trial registration number for the SELECT trial is NCT03574597.
This is a developing story. Share your thoughts and questions in the comments below.