When a stroke damages part of the brain, the immediate focus is often on loss—of movement, speech, or memory. But emerging research reveals a surprising counterpoint: while the injured hemisphere may show signs of accelerated aging, the unaffected side can appear biologically younger, as if the brain is tapping into a hidden reserve of resilience. This phenomenon, observed in neuroimaging studies of stroke survivors, suggests the brain’s capacity for reorganization extends beyond simple compensation—it may actively rejuvenate healthy regions to support recovery.
The idea challenges long-held assumptions about neural aging after injury. Traditionally, brain damage was thought to trigger a uniform decline, with surrounding tissue succumbing to secondary injury or maladaptive changes. Yet advanced MRI analyses now show that in some patients, the contralateral hemisphere exhibits structural and metabolic features associated with younger brain tissue—increased gray matter volume, enhanced connectivity, and reduced markers of cellular aging. These changes correlate with better functional outcomes, hinting that the brain doesn’t just adapt—it may actively renew.
To understand what “brain rejuvenation” means researchers appear beyond chronological age to biological markers visible in neuroimaging. One key measure is brain age gap—the difference between a person’s actual age and the age estimated from their brain scan using machine learning models. A negative gap (where the brain looks younger than expected) has been linked to cognitive reserve, physical fitness, and genetic factors. In stroke studies, a younger-appearing unaffected hemisphere has been associated with greater neuroplasticity and improved rehabilitation response.
This pattern isn’t universal. Not all stroke survivors show this contralateral youthfulness, and the mechanisms remain under investigation. However, the observation opens a new avenue: could we harness this intrinsic capacity to boost recovery? Scientists are exploring whether interventions like aerobic exercise, cognitive training, or neuromodulation might amplify this natural rejuvenation signal, turning a passive observation into an active therapeutic strategy.
The Science Behind the Scan: What Does a “Younger-Looking” Brain Mean?
When scientists say a brain region “looks younger” on a scan, they’re not referring to appearance in the literal sense. Instead, they apply predictive models trained on thousands of healthy brain images to estimate biological age based on patterns of atrophy, white matter integrity, and cortical thickness. These models, developed by groups such as those at the Medical Research Council Laboratory of Molecular Biology and validated in large cohorts like UK Biobank, can predict chronological age with remarkable accuracy—often within a few years.
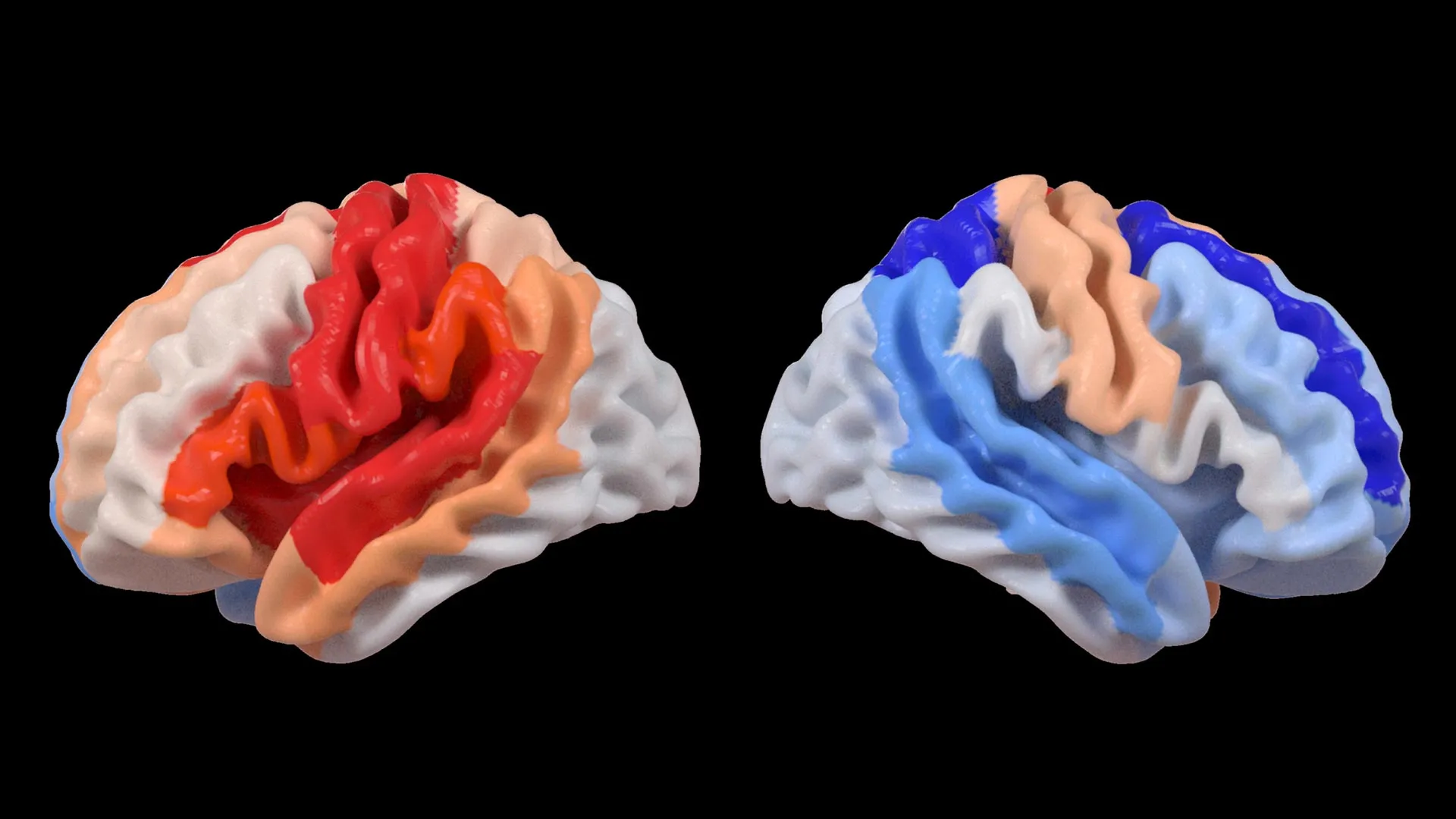
In stroke research, applying these models to survivors has revealed a striking dissociation. A 2023 study published in Neurology analyzed longitudinal MRI data from 512 ischemic stroke patients and found that while the infarct-affected hemisphere showed an average brain age gap of +2.1 years (indicating accelerated aging), the homologous region in the opposite hemisphere showed a gap of -1.4 years—suggesting a younger biological profile. The effect was most pronounced in patients under 70 with moderate severity strokes and was linked to better scores on motor function tests at six-month follow-up.
Importantly, this doesn’t mean the unaffected side is literally reversing time. Rather, it may reflect heightened neuroplasticity—increased synaptic density, glial support, or vascular efficiency—that mimics the structural profile of a younger brain. Animal studies support this: after induced stroke, rodents show upregulation of growth factors like BDNF and VEGF in the contralateral cortex, promoting dendritic branching and angiogenesis. Whether similar molecular cascades occur in humans is an active area of research, with PET and MR spectroscopy studies beginning to probe metabolic signatures of recovery.
Why Does This Happen? The Brain’s Bidirectional Response to Injury
The brain’s reaction to stroke isn’t passive damage—it’s a dynamic, body-wide response involving inflammation, hormonal shifts, and neural rewiring. In the acute phase, the injured area releases signals that trigger both harmful (excitotoxicity, inflammation) and helpful (cleanup, repair) processes. Over days to weeks, the focus shifts to adaptation: surviving neurons form new connections, dormant pathways are unmasked, and homologous regions in the opposite hemisphere seize on added responsibility.
This contralateral recruitment is well-documented in motor recovery. For example, after a left-hemisphere stroke affecting right-hand movement, fMRI often shows increased activation in the right motor cortex during attempted hand tasks—a pattern associated with better outcomes in some patients. What’s newer is the suggestion that this isn’t just functional overlay but structural enhancement. The unaffected side may not only work harder—it may physically remodel itself to support the increased load.
Factors influencing this response include age, genetics, pre-stroke cognitive reserve, and rehabilitation intensity. Younger patients and those with higher education or bilingualism often show stronger contralateral adaptations, possibly reflecting greater neural reserve. Conversely, vascular risk factors like hypertension or diabetes may blunt this response, contributing to the variability seen across populations.
What This Means for Recovery and Rehabilitation
If the brain can selectively rejuvenate regions to aid recovery, rehabilitation strategies might be optimized to nurture this process. Current guidelines emphasize early, intensive, task-specific therapy—but could timing, type, or intensity be adjusted to align with the brain’s natural plasticity windows? Some researchers propose that combining physical therapy with transcranial direct current stimulation (tDCS) targeting the unaffected hemisphere might enhance, rather than inhibit, its adaptive role—a nuance still debated in the field.
Lifestyle factors also appear influential. Observational data from the American Heart Association suggest that stroke survivors who engage in regular aerobic activity, maintain a Mediterranean-style diet, and manage stress show better long-term cognitive and functional trajectories—potentially by supporting the biological conditions under which contralateral rejuvenation occurs.
Still, caution is warranted. An overly active or maladaptive contralateral hemisphere can sometimes hinder recovery—for instance, by interfering with the re-engagement of the injured side through interhemispheric inhibition. Balance is key: the goal isn’t to maximize activity in the healthy side, but to promote efficient, coordinated network function across both hemispheres.
The Road Ahead: From Observation to Intervention
While the link between a younger-appearing contralateral hemisphere and better outcomes is compelling, causality remains to be proven. Does the brain rejuvenate to support recovery, or do patients with greater innate resilience simply show both better recovery and a younger brain signature? Longitudinal studies tracking changes before and after stroke—though rare due to the unpredictability of the event—are beginning to shed light. The National Institute of Neurological Disorders and Stroke supports several such cohorts, including SPRINT-MIND and sub-studies of the ARIC trial, which now include neuroimaging biomarkers of brain aging.
Future directions include multimodal imaging—combining MRI with PET to assess microglial activation, amyloid burden, or neurotransmitter systems—to distinguish true rejuvenation from mere compensation. Trials are also testing whether drugs that promote neuroplasticity (such as SSRIs or statins, which have pleiotropic effects beyond their primary use) can amplify the brain’s intrinsic repair signals when paired with rehabilitation.
For now, the message is one of cautious optimism. The brain’s response to injury is far more complex—and potentially more regenerative—than once believed. By recognizing that healing isn’t just about salvaging the damaged, but also about nurturing the resilient, we may open new pathways to recovery that work with, rather than against, the brain’s own intelligence.
Understanding how stroke reshapes the brain—both its wounds and its hidden strengths—remains a vital frontier in neurology. As imaging tools grow sharper and longitudinal data deeper, we may yet learn to read not just the scars of injury, but the quiet signs of renewal.
If you or someone you know is navigating life after stroke, consider discussing rehabilitation options and brain health strategies with a neurologist or physiatrist. Staying informed empowers better decisions—and every step toward recovery counts.
Share your thoughts or experiences in the comments below, and help others by passing this along to anyone who might find it useful.