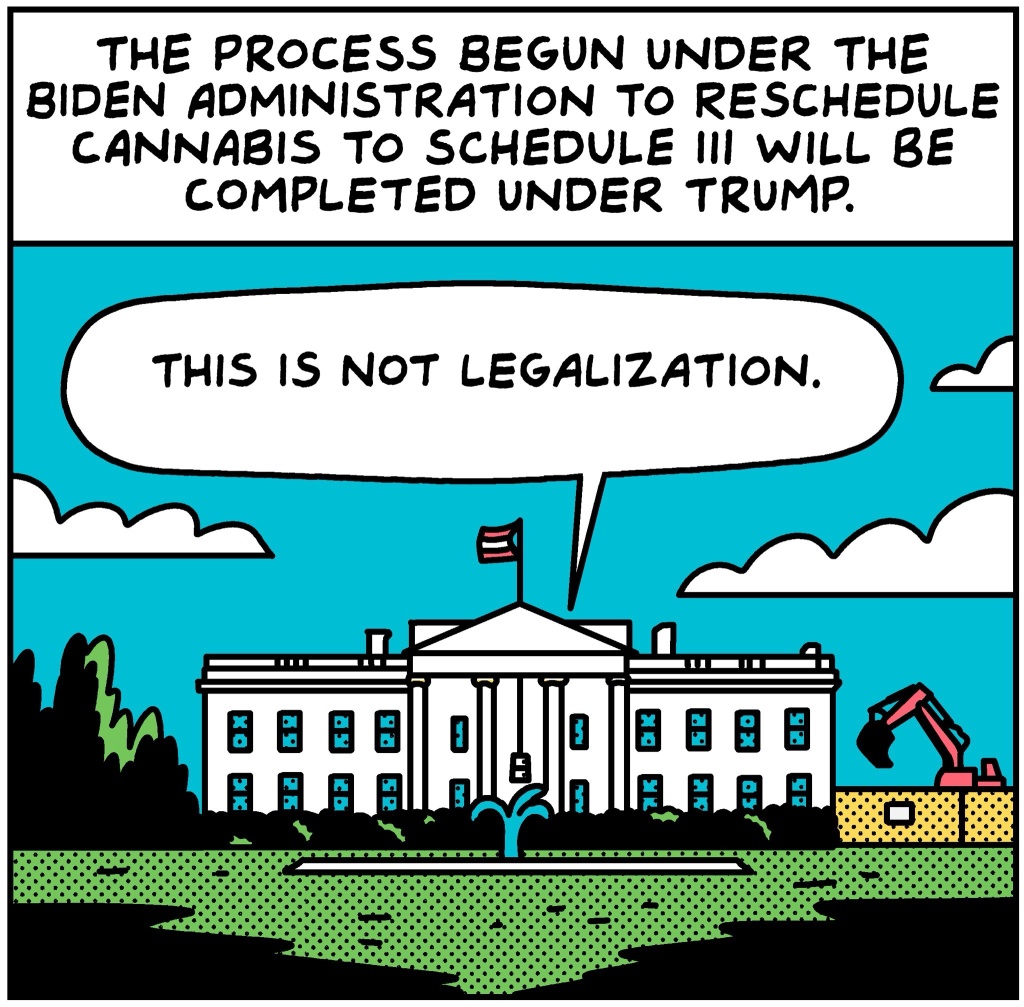
The U.S. Department of Justice announced on Thursday, April 23, 2026, that We see reclassifying marijuana from Schedule I to Schedule III under the Controlled Substances Act, a move intended to facilitate medical research and expand patient access to cannabis-based treatments.
The decision, confirmed by Acting Attorney General Todd Blanche, follows a presidential directive signed by former President Donald Trump on December 18, 2025, aimed at encouraging scientific study of cannabis’s therapeutic potential. While the reclassification does not federally legalize marijuana, it acknowledges a lower potential for abuse and places it in the same category as substances like ketamine and certain codeine-containing medications.
According to the Justice Department, approximately three-quarters of Americans now live in states where cannabis is legal for medical or recreational leverage, yet federal prohibition has long hindered clinical research and created regulatory barriers for businesses operating in state-legal markets.
The rescheduling triggers an accelerated administrative review process beginning June 29, 2026, to evaluate whether marijuana should be rescheduled further or potentially removed from federal control altogether, beyond its current medical-use focus.
“These measures will enable more targeted and rigorous research on the safety and efficacy of marijuana, broaden patient access to treatments and allow healthcare providers to make better-informed medical decisions,” Blanche stated in a press release distributed by the Associated Press.
The shift could have significant economic implications for the cannabis industry, reducing tax burdens under Section 280E of the Internal Revenue Code and easing banking restrictions that have long plagued state-licensed operators.
Former President Joe Biden had previously advocated for federal decriminalization of marijuana during his term, but legislative efforts stalled in Congress. The Trump administration’s approach, by contrast, emphasizes administrative action through the Drug Enforcement Administration (DEA) and the Department of Health and Human Services (HHS), which jointly recommended the rescheduling based on a review of scientific evidence.
Under the Controlled Substances Act, Schedule I substances are defined as having a high potential for abuse, no currently accepted medical use, and a lack of accepted safety for use under medical supervision. Schedule III drugs, by contrast, are recognized to have a moderate to low potential for physical and psychological dependence, with accepted medical applications.
The move aligns with growing public support for cannabis reform. A 2025 Pew Research Center survey found that 88% of U.S. Adults support legalizing marijuana for medical or recreational use, with 59% favoring both medical and legal recreational access.
Medical cannabis is currently used to treat conditions such as chronic pain, epilepsy, multiple sclerosis-related spasticity, and nausea induced by chemotherapy. The reclassification may streamline FDA approval pathways for cannabis-derived pharmaceuticals and reduce barriers for investigators seeking to conduct clinical trials.
Industry groups, including the U.S. Cannabis Council and the National Organization for the Reform of Marijuana Laws (NORML), welcomed the change as a step toward resolving the conflict between state and federal law, though they urged Congress to pass comprehensive reform legislation to fully deschedule marijuana.
Legal experts note that while rescheduling removes some federal penalties and research obstacles, it does not expunge prior convictions, allow interstate commerce, or permit possession on federal lands. Ongoing advocacy continues to focus on the Marijuana Opportunity Reinvestment and Expungement (MORE) Act and related bills aimed at addressing social equity in cannabis licensing.
The next key deadline in the process is June 29, 2026, when the accelerated review for broader rescheduling is set to begin. Stakeholders await further guidance from the DEA and HHS on the scientific and public health evaluation that will inform any future scheduling decisions.
For ongoing updates, readers can refer to the Federal Register notices published by the Drug Enforcement Administration or visit the official website of the U.S. Department of Justice.
We invite our global audience to share perspectives on how this policy shift may influence international drug reform efforts. Join the conversation in the comments below and share this article to help foster informed discussion.