The fight against Alzheimer’s disease is entering a transformative era, shifting from merely managing symptoms to actively reprogramming the brain’s internal defense mechanisms. For decades, the medical community has viewed the accumulation of amyloid-beta plaques as the primary driver of cognitive decline. However, the most recent breakthroughs focus not just on removing these plaques, but on the cleaning crew
responsible for the job: the microglia.
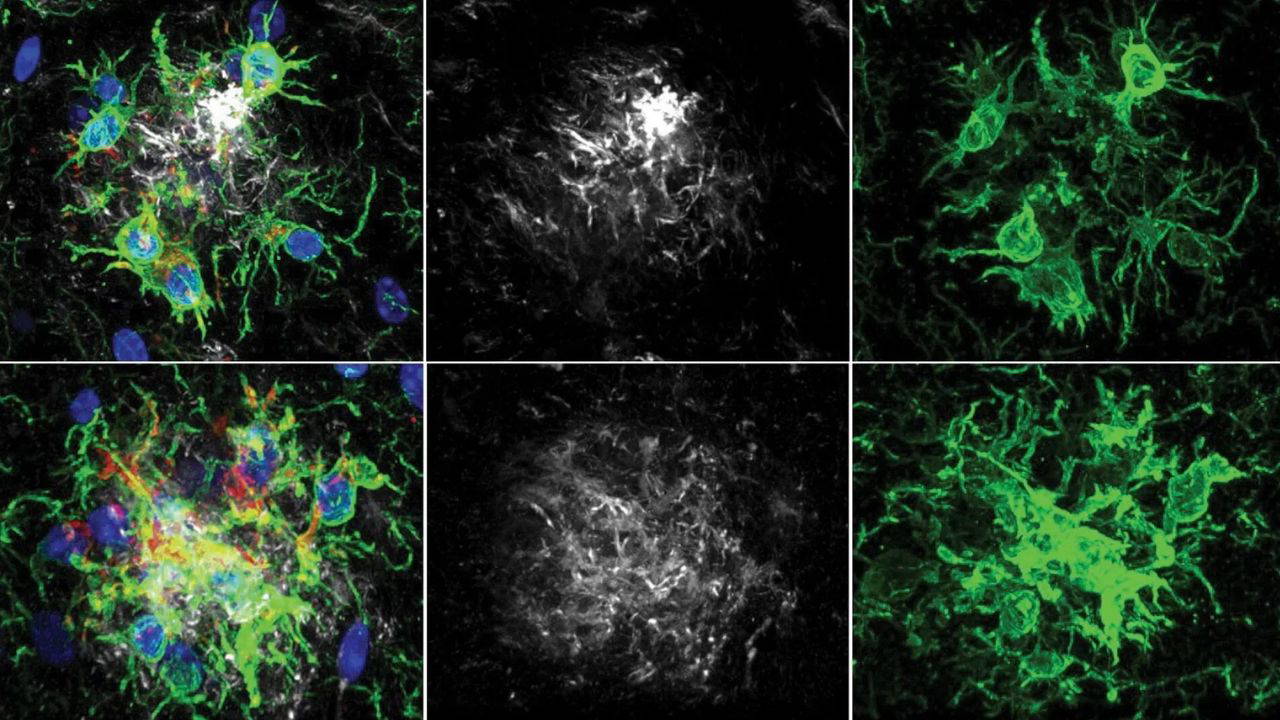
Microglia are the resident immune cells of the central nervous system, acting as the brain’s primary waste-management system. In a healthy brain, these cells identify and clear metabolic debris and toxic proteins. In patients with Alzheimer’s, however, microglia often become dysfunctional or “exhausted,” losing their ability to clear amyloid-beta and instead triggering chronic inflammation that further damages neurons. The emergence of microglia reprogramming as a therapeutic strategy represents a pivotal shift in neuropharmacology.
As Editor of Health at World Today Journal and a physician trained at Charité – Universitätsmedizin Berlin, I have tracked the evolution of these therapies from early monoclonal antibodies to the current focus on cellular engineering. The goal is no longer just to introduce a drug that binds to a plaque, but to “re-educate” the brain’s own immune cells to recognize and eliminate pathology more efficiently.
Reprogramming the Brain’s Immune Response
Recent research indicates that the efficacy of modern Alzheimer’s treatments depends heavily on how they interact with microglia. For instance, the therapeutic agent lecanemab does not simply dissolve plaques on its own; rather, it induces an amyloid-clearing program
within the microglia. According to a study published in Nature Neuroscience, lecanemab activates microglial effector functions, effectively triggering the cells to engulf and remove amyloid-beta pathology.
Further insights published in March 2026 reveal that this process is mediated by a specific part of the antibody known as the Fc fragment. This fragment acts as a molecular trigger, prompting microglia to transition from a dormant or dysfunctional state into an active, phagocytic state capable of clearing harmful plaques. This discovery is critical because it allows scientists to design future therapies that can stimulate the “cleaning” function of microglia without triggering the harmful inflammatory responses that can lead to brain swelling or microhemorrhages.
Beyond antibodies, researchers are exploring “cellular couriers.” In April 2025, scientists at the University of California, Irvine, demonstrated a platform for delivering disease-fighting proteins by engineering human microglia. These engineered cells are designed to respond to specific brain signals, allowing for a targeted, brain-wide delivery of therapeutic agents that can be reprogrammed to tackle various neurological disorders ScienceDaily.
The Role of Metabolic and Epigenetic Shifts
The failure of microglia in Alzheimer’s is often rooted in metabolic dysfunction. Recent evidence suggests that the cells undergo a form of “glycolytic reprogramming,” where their energy production shifts in a way that impairs their ability to perform phagocytosis (the process of eating debris). If the microglia cannot produce energy efficiently, they cannot clear the plaques, regardless of whether the plaques are “tagged” for removal.
Epigenetic factors as well play a significant role. Research published in March 2026 in the Journal of Neuroinflammation highlights how the ApoE4 variant—the strongest genetic risk factor for late-onset Alzheimer’s—drives lipid dysregulation in microglia through epigenetic reprogramming. This suggests that “fixing” the cleaning crew may require a combination of metabolic support and epigenetic modulation to restore their original function.
A New Diagnostic Paradigm: The Rise of Blood Tests
While the focus on microglia addresses how to treat the disease, a parallel revolution is occurring in how we detect it. For years, a definitive Alzheimer’s diagnosis required invasive cerebrospinal fluid (CSF) punctures or expensive PET scans. That paradigm shifted significantly in 2025.
On May 16, 2025, the U.S. Food and Drug Administration (FDA) cleared for marketing the first in vitro diagnostic device that tests blood to aid in diagnosing Alzheimer’s disease FDA. Specifically, the Lumipulse G pTau217/ß-Amyloid 1-42 Plasma Ratio test allows for the early detection of amyloid plaques in adult patients aged 55 and older.
This advancement is not merely a convenience; it is a clinical necessity. For microglia-reprogramming therapies to be effective, they must be administered before widespread neuronal death occurs. The ability to screen high-risk populations via a simple blood draw means patients can be identified in the “prodromal” or preclinical stage, where the brain’s immune system is still responsive enough to be reprogrammed.
Comparing Traditional vs. Modern Diagnostic Approaches
| Method | Procedure | Invasiveness | Primary Marker |
|---|---|---|---|
| Traditional Clinical | Cognitive tests/History | Non-invasive | Symptomatic behavior |
| CSF Analysis | Lumbar Puncture | High | Tau and Amyloid levels |
| PET Imaging | Radiotracer Injection | Moderate | Amyloid/Tau plaque density |
| Modern Blood Test | Venous Blood Draw | Low | pTau217/ß-Amyloid 1-42 ratio |
What In other words for Patients and Caregivers
The convergence of early detection via blood tests and the ability to modulate microglia creates a new “window of opportunity.” In the past, by the time a patient exhibited significant memory loss, the microglia were often too depleted or dysfunctional to be effectively reversed. With the ability to detect amyloid pathology years before symptoms appear, the goal is to maintain cognitive health by keeping the brain’s “cleaning crew” active throughout the aging process.
However, it is important to manage expectations. Reprogramming microglia is a complex biological feat. While preclinical mouse models have shown the ability to ameliorate pathology through microglia replacement and modulation, translating these results to human patients requires rigorous safety testing. The risk of “over-activating” the immune system can lead to neuroinflammation, which may accelerate cognitive decline rather than stop it.
Key Takeaways for the Global Health Community
- Microglia as Targets: Treatment is shifting from targeting plaques (the “trash”) to targeting microglia (the “trash collectors”).
- Mechanism of Action: New drugs like lecanemab work by activating a specific amyloid-clearing program in the brain’s immune cells.
- Early Detection: FDA-cleared blood tests for pTau217 now allow for earlier intervention, which is critical for the success of cellular therapies.
- Genetic Influence: The ApoE4 gene doesn’t just increase plaque risk; it actively disables the microglia’s ability to clear those plaques.
The Path Forward
The next critical checkpoint in this research will be the results of ongoing human trials focusing on “next-generation” microglia modulators—drugs designed specifically to target the Fc fragment or metabolic pathways without the side effects associated with first-generation antibodies. We are also awaiting expanded global rollout of blood-based biomarkers, which will likely integrate into routine geriatric care in the coming years.

As we move toward a future of “precision neurology,” the focus remains on the synergy between early diagnosis and cellular reprogramming. We are no longer just observing the decline; we are learning how to restart the brain’s own healing processes.
Do you or a loved one have experience with the new generation of Alzheimer’s diagnostics or treatments? Share your thoughts and questions in the comments below to join our global conversation on brain health.