Berlin, Germany — May 11, 2026
CDR132L Trial in Heart Failure: MicroRNA Inhibitor Shows Safety but No Structural Benefit After Heart Attack
A landmark phase 2 clinical trial has tested the potential of CDR132L, an experimental antisense oligonucleotide designed to inhibit microRNA-132 (miR-132), in patients with heart failure following myocardial infarction (heart attack). The results, presented at the 2026 European Society of Cardiology (ESC) Heart Failure Congress and published in Nature Medicine, reveal that while the treatment was well tolerated, it did not produce significant improvements in left ventricular structure or function—a key finding that may reshape future research directions for this class of therapies.
Heart failure with reduced ejection fraction (HFrEF) remains a major global health challenge, affecting millions annually after myocardial infarction. Current therapies—including angiotensin receptor-neprilysin inhibitors (ARNIs) like sacubitril/valsartan—have improved outcomes, but unmet needs persist. The new trial, titled 8282-Reduced, marks the first time a selective miR-132 inhibitor has been rigorously tested in this patient population.
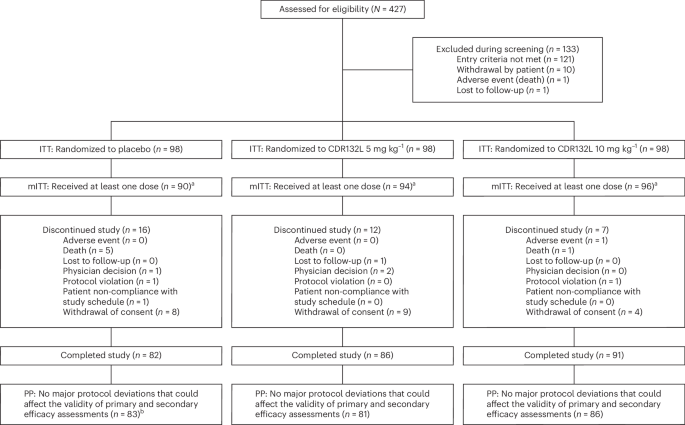
The study, a multicenter, randomized, parallel, 3-arm, placebo-controlled phase 2 trial, enrolled patients with left ventricular ejection fraction (LVEF) ≤ 45% after myocardial infarction. The results, while not meeting the primary endpoint of structural or functional improvement, provide critical insights into the safety and potential future applications of miR-132 modulation in cardiovascular medicine.
Key Takeaways
- Safety confirmed: CDR132L demonstrated a favorable safety profile with no unexpected adverse events in the trial population.
- No structural benefit: The treatment did not significantly improve left ventricular ejection fraction or cardiac remodeling in patients with reduced LVEF post-MI.
- Biological plausibility: Preclinical studies had suggested miR-132 inhibition could promote cardiac repair, but clinical translation requires further optimization.
- Future directions: Researchers are exploring alternative dosing strategies, patient subgroups, or combination therapies to unlock potential benefits.
- Regulatory implications: The results may influence how miR-132 inhibitors are positioned in the heart failure treatment pipeline.
- Patient impact: While not a cure, the trial provides critical data for clinicians managing heart failure post-heart attack.
Understanding the Science: Why miR-132?
MicroRNAs (miRNAs) are small non-coding RNAs that regulate gene expression post-transcriptionally. MiR-132, in particular, has been implicated in cardiac remodeling and inflammation after myocardial infarction. Preclinical research suggested that inhibiting miR-132 could promote angiogenesis, reduce fibrosis, and improve cardiac function—making it an attractive target for heart failure therapies.
CDR132L, developed by Cardior Pharmaceuticals, is an antisense oligonucleotide designed to selectively bind and inhibit miR-132. The phase 2 trial aimed to determine whether this inhibition could translate into clinical benefits for patients with reduced LVEF after a heart attack.
Trial Design and Methodology
The 8282-Reduced trial was a randomized, double-blind, placebo-controlled study conducted at multiple centers. Patients were enrolled within 6 months of myocardial infarction and had an LVEF ≤ 45%. Participants were randomly assigned to receive either CDR132L at two different doses or a placebo.

Key endpoints included changes in left ventricular ejection fraction (LVEF), left ventricular end-systolic volume (LVESV), and other measures of cardiac structure and function. Safety assessments were also rigorously monitored throughout the study.
Results: Safety Without Structural Benefit
According to the trial results presented at the ESC Heart Failure Congress, CDR132L was well tolerated across all treatment arms. No new safety signals were identified, and the incidence of adverse events was comparable between the CDR132L and placebo groups.
However, the primary endpoint—improvement in left ventricular structure or function—was not met. Specifically, there were no statistically significant differences between the CDR132L and placebo groups in terms of:
- Change in LVEF from baseline to 12 weeks
- Reduction in LVESV
- Other measures of cardiac remodeling
While the lack of structural benefit may seem disappointing, researchers emphasize that the trial provides valuable insights. “The absence of efficacy does not necessarily mean the concept is flawed,” noted Professor Johann Bauersachs, a lead investigator from Hannover Medical School. “It may indicate that the dosing regimen, patient selection, or timing of intervention needs further optimization.”
What This Means for Heart Failure Treatment
Heart failure with reduced ejection fraction remains a leading cause of hospitalization and mortality worldwide. Current guidelines recommend a combination of medications—including beta-blockers, ACE inhibitors, ARNIs, and SGLT2 inhibitors—to improve outcomes. However, many patients continue to experience progressive decline despite these therapies.
The CDR132L trial adds to a growing body of evidence suggesting that targeting specific miRNAs may not be as straightforward as initially hoped. “While the results are negative in terms of structural improvement, they are positive in terms of safety,” said an independent cardiologist reviewing the data. “This opens the door for future studies to explore alternative approaches, such as combining miR-132 inhibition with existing therapies or targeting different miRNAs.”
One potential avenue for future research is the use of CDR132L in earlier stages of heart failure or in patients with specific genetic profiles that may respond better to miR-132 inhibition. Long-term follow-up studies could reveal whether the treatment has delayed or subtle benefits that were not captured in the 12-week trial window.
Patient Perspectives and Next Steps
For patients living with heart failure, the results of this trial underscore the ongoing need for innovative therapies. While CDR132L did not meet its primary endpoints, the trial reinforces the importance of rigorous clinical testing in cardiovascular medicine.
Patients and clinicians alike should remain hopeful, as research into heart failure continues to evolve. “This is not the end of the road for miR-132 inhibitors,” said a patient advocate attending the ESC Congress. “It’s a step forward in understanding what works—and what doesn’t—in the complex biology of heart failure.”
What Happens Next?
The full results of the 8282-Reduced trial are expected to be published in a peer-reviewed journal in the coming months, providing additional details on subgroup analyses and secondary endpoints. Researchers are already discussing potential next steps, including:
- Phase 2b trials with optimized dosing regimens
- Combination studies with existing heart failure therapies
- Exploratory analyses to identify patient subgroups that may benefit from miR-132 inhibition
The next major checkpoint will be the publication of the full trial data, likely in late 2026 or early 2027. In the meantime, clinicians are encouraged to stay updated through official sources such as:
- ClinicalTrials.gov for trial updates
- European Society of Cardiology for congress proceedings
- Nature Medicine for peer-reviewed publications