A new combination therapy is showing promising results in the fight against one of the most aggressive forms of cancer. Recent data from a randomized phase 2 trial indicates that adding elraglusib and chemotherapy in metastatic pancreatic ductal adenocarcinoma (PDAC) significantly extends overall survival for patients who have not previously received systemic therapy.
The findings, presented at the ASCO Gastrointestinal Cancers Symposium 2026, suggest that the addition of elraglusib—a novel compact-molecule inhibitor—to the standard chemotherapy regimen of gemcitabine and nab-paclitaxel (GnP) provides a meaningful survival benefit. For patients facing a diagnosis of metastatic pancreatic cancer, these results offer a potential new pathway for improving clinical outcomes in a disease known for its limited treatment options.
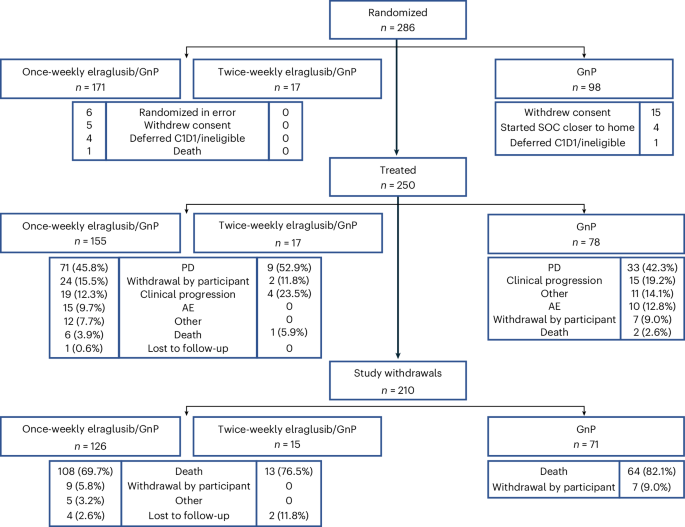
The study, known as Part 3B of the phase 2 1801 trial (NCT03678883), focused on patients with metastatic PDAC who were at least 18 years old and had measurable disease. Notably, the trial included a broad range of patients, with no minimum life expectancy requirement and no exclusion for those with high carbohydrate antigen (CA) 19-9 levels or low albumin, reflecting a real-world patient population according to data presented at the ASCO Gastrointestinal Cancers Symposium 2026.
The Role of GSK-3β Inhibition in Pancreatic Cancer
Elraglusib, also identified as 9-ING-41, functions as a GSK-3β inhibitor. Specifically, It’s a cell-permeable ATP-competitive inhibitor of glycogen synthase kinase-3β via PubMed. By targeting this specific enzyme, the drug aims to disrupt the biological pathways that allow pancreatic cancer cells to survive and resist standard chemotherapy.
When used alone, chemotherapy such as gemcitabine and nab-paclitaxel is a standard approach for treating metastatic PDAC. However, the addition of a GSK-3β inhibitor like elraglusib is designed to enhance the efficacy of these agents, potentially making the tumor more susceptible to the cytotoxic effects of the chemotherapy.
Breaking Down the Trial Results
The analysis for the 1801 Part 3B trial included a total of 233 patients. Of these, 155 patients received either weekly or twice-weekly elraglusib (at a dose of 9.3 mg/kg) combined with a 28-day cycle of gemcitabine and nab-paclitaxel. The control group consisted of 78 patients who received gemcitabine and nab-paclitaxel alone as reported by Oncology Nurse Advisor.
The results demonstrated a clear divergence in survival rates between the two groups. As of the data cutoff on November 22, 2025, the study met its primary endpoint. The 12-month overall survival rate was 44.4% for those treated with elraglusib, compared to 22.3% for those receiving chemotherapy alone per the ASCO Gastrointestinal Cancers Symposium 2026 presentation.
the median overall survival (OS) for patients in the elraglusib group was 10.1 months. This represents a significant increase over the survival observed in the group receiving only gemcitabine and nab-paclitaxel according to trial data.
Key Trial Statistics
| Metric | Elraglusib + GnP | GnP Alone |
|---|---|---|
| Number of Patients | 155 | 78 |
| 12-Month Overall Survival Rate | 44.4% | 22.3% |
| Median Overall Survival | 10.1 months | Lower (Not specified) |
| Indicate Age of Participants | 65.1 years | 66.2 years |
Patient Demographics and Study Design
The trial was carefully balanced to ensure that the results were attributable to the medication rather than baseline patient differences. The mean age of patients was 65.1 years in the elraglusib group and 66.2 years in the control group. Gender distribution was also similar, with just over half of the patients in both arms being male according to the study findings.
Researchers confirmed that baseline CA 19-9 levels, the sites of the disease, and the overall performance status of the patients were comparable between the two arms of the study. This consistency strengthens the conclusion that the improved survival rates were linked to the addition of the GSK-3β inhibitor.
Expert Analysis and Future Implications
Dr. Devalingam Mahalingam, MD, PhD, of the Robert H. Lurie Comprehensive Cancer Center at Northwestern University, who presented the study, emphasized the importance of these findings. “Given the context that What we have is a randomized clinical trial, our study had a survival meaningful benefit and lays the foundation for GSK-3beta inhibition in subsequent studies in pancreatic cancer, along with other tumor types,” Dr. Mahalingam stated via Oncology Nurse Advisor.

The success of this phase 2 trial is a critical step toward potentially changing the standard of care for metastatic PDAC. Because pancreatic cancer is often diagnosed at an advanced stage and is notoriously resistant to many therapies, any combination that significantly increases the 12-month survival rate is viewed as a major development by the oncology community.
The study demonstrates that targeting GSK-3β can potentially sensitize pancreatic tumors to chemotherapy, providing a biological rationale for further exploration of this inhibitor in other hard-to-treat malignancies.
Key Takeaways for Patients and Caregivers
- New Combination: The trial tested the addition of elraglusib (a GSK-3β inhibitor) to the standard gemcitabine and nab-paclitaxel chemotherapy.
- Survival Boost: The 12-month survival rate nearly doubled from 22.3% (chemotherapy alone) to 44.4% (combination therapy) per trial results.
- Median Survival: Patients receiving the combination therapy saw a median overall survival of 10.1 months.
- Broad Eligibility: The study included patients regardless of their albumin levels or CA 19-9 markers, suggesting the benefit may be applicable to a wide range of metastatic PDAC patients.
Although these results are encouraging, this was a phase 2 trial. Further large-scale phase 3 trials are typically required to confirm these benefits and establish safety profiles before a treatment becomes widely available as a standard clinical practice.
The medical community now looks toward subsequent studies to determine if GSK-3β inhibition can be effectively applied to other tumor types beyond pancreatic cancer, as suggested by the study’s lead presenter.
For the latest updates on this trial and other medical innovations in oncology, patients and healthcare providers are encouraged to monitor official announcements from the ASCO (American Society of Clinical Oncology) and clinical trial registries such as ClinicalTrials.gov (NCT03678883).
We welcome your thoughts and questions on these developments in cancer research. Please share this article with others who may find this information valuable.