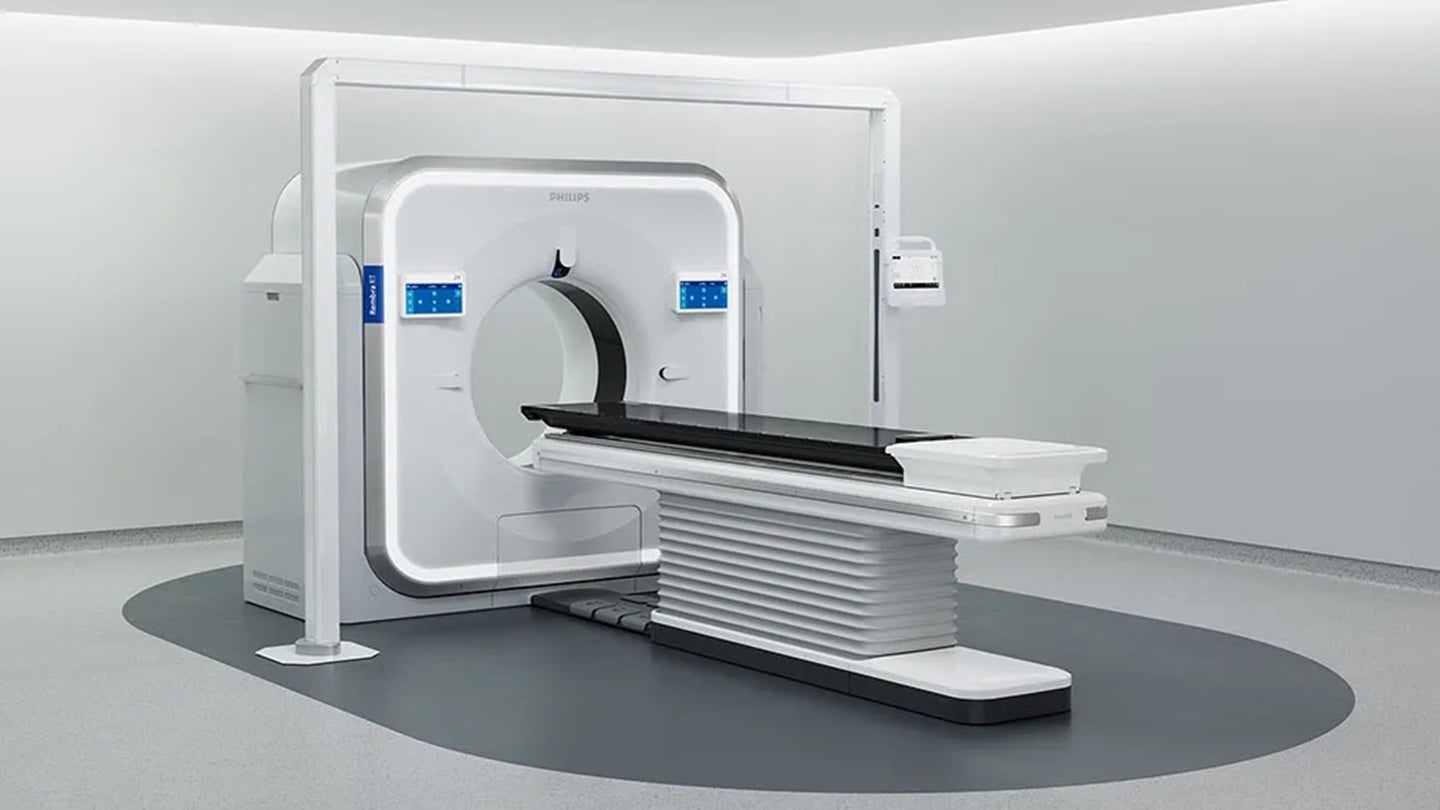
Royal Philips has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Rembra platform of scanning systems, marking a significant advancement in medical imaging technology. The clearance, announced on April 22, 2026, covers the Rembra CT, Rembra RT, and Areta RT systems, enabling broader access to faster and more precise computed tomography (CT) imaging across frontline care and cancer treatment planning settings.
The FDA’s decision allows Philips to expand the availability of its integrated imaging solutions designed to meet rising demands for speed, efficiency, and high-quality diagnostic capabilities. According to the company’s official press release, the Rembra platform supports healthcare providers in responding to increasing imaging volumes and clinical complexity by improving workflow efficiency without compromising diagnostic confidence.
Dan Xu, Business Leader of CT at Philips, emphasized the platform’s role in modernizing clinical expectations: “As healthcare systems manage increasing demand and complexity, imaging plays a critical role in enabling timely and informed clinical decisions. With the Rembra platform, we are redefining what clinicians can expect from CT, combining speed, scalability, and precision to expand access to high-quality imaging while supporting confident diagnosis and highly accurate treatment planning.”
The Rembra CT system features an 85 cm bore and is engineered for ultra-fast, high-throughput imaging in intensive clinical environments such as emergency departments and critical care units. It supports up to 270 examinations per day, incorporating advanced acquisition and reconstruction capabilities aimed at facilitating timely decision-making. These technical specifications are designed to maintain image quality even under high-volume conditions.
In radiation therapy applications, the Rembra RT and Areta RT systems extend the platform’s advantages into treatment planning. These systems offer an 85 cm extended field of view and next-generation 4DCT imaging capabilities, which are specifically developed to improve tumor targeting accuracy while minimizing radiation exposure to surrounding healthy tissue. This precision supports more personalized and effective cancer care strategies.
The integration of diagnostic radiology and radiation therapy workflows represents a key innovation of the Rembra platform. By enabling a seamless transition from rapid assessment in acute settings to detailed treatment planning, the system promotes a more coordinated approach to patient care. This connectivity is particularly valuable in oncology, where timely and accurate imaging directly influences therapeutic outcomes.
Industry analysts note that the FDA clearance comes at a time when healthcare systems globally are under pressure to deliver faster diagnostics amid rising patient volumes and staffing challenges. Technologies that enhance throughput without sacrificing diagnostic accuracy are increasingly seen as essential tools in modern healthcare delivery.
Philips states that the Rembra platform builds on its long-standing expertise in imaging innovation, incorporating feedback from clinicians and healthcare administrators to address real-world operational challenges. The system’s design prioritizes ease of use, reliability, and scalability across diverse clinical settings, from community hospitals to large academic medical centers.
As of the announcement date, no public details have been released regarding pricing, specific installation timelines, or regional rollout plans for the Rembra platform. Interested parties are encouraged to consult Philips’ official investor relations page or regulatory filings for future updates on commercial availability and market strategy.
The FDA’s 510(k) clearance pathway requires manufacturers to demonstrate that a new device is substantially equivalent to a legally marketed predicate device. This regulatory route is commonly used for moderate-risk medical devices and allows for relatively swift market entry following successful review.
Moving forward, stakeholders in medical imaging, oncology, and hospital administration will likely monitor the adoption and clinical impact of the Rembra platform. Real-world performance data, user feedback, and comparative effectiveness studies will be important factors in assessing its long-term value to healthcare systems.
For ongoing updates on regulatory approvals, technological advancements, and implementation insights related to the Rembra platform and similar innovations in medical imaging, readers can refer to trusted sources such as the U.S. FDA’s medical device database, peer-reviewed journals in radiology and oncology, and official communications from Royal Philips.
We invite healthcare professionals, technologists, and policymakers to share their perspectives on how integrated imaging platforms like Rembra are shaping the future of diagnostics and treatment planning. Join the conversation by commenting below or sharing this article with your network.