Recent research continues to illuminate the complex relationship between the gut microbiome and Parkinson’s disease, offering new insights into how microbial changes may precede and potentially influence neurodegenerative processes. A study published in Nature Medicine in April 2026 analyzed gut microbial profiles across a spectrum of individuals — from those with no known risk to genetically predisposed carriers and clinically diagnosed patients — revealing a progressive pattern of dysbiosis that correlates with disease proximity.
The findings suggest that alterations in gut bacteria are not merely a consequence of Parkinson’s disease but may represent an early biological signature detectable years before motor symptoms emerge. This evolving understanding positions the microbiome as a potential window into preclinical disease stages, opening avenues for earlier detection and preventive strategies. As scientists refine these microbial markers, the implications for public health and clinical practice grow increasingly significant.
Parkinson’s disease affects over 10 million people worldwide, according to the World Health Organization, with prevalence rising due to aging populations. Even as genetic factors like mutations in the LRRK2 or GBA genes increase risk, most cases involve a mix of inherited susceptibility and environmental triggers. The gut-brain axis — a bidirectional communication network linking gastrointestinal function and central nervous system activity — has emerged as a key area of interest in understanding how distant bodily systems might influence brain health.
Tracking Microbial Shifts Across Disease Stages
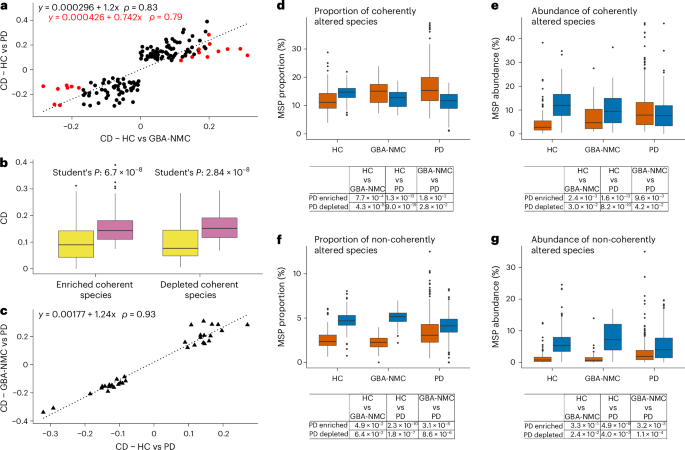
In the 2026 study, researchers sequenced stool samples from three cohorts: healthy controls with no family history, individuals carrying high-risk genetic variants (such as GBA mutations) but without symptoms, and patients with clinically confirmed Parkinson’s disease. Analysis revealed a gradient of microbial changes, where certain beneficial bacteria decreased and pro-inflammatory taxa increased in stepwise fashion across the groups.
Notably, even genetically at-risk individuals who remained asymptomatic showed measurable deviations in microbiome composition compared to healthy controls. These included reduced levels of Faecalibacterium prausnitzii, a bacterium known for producing anti-inflammatory short-chain fatty acids, and elevated abundances of Ruminococcus gnavus, which has been associated with gut barrier disruption in other neurodegenerative conditions.
The degree of dysbiosis — measured through diversity indices and taxonomic shifts — correlated closely with proximity to clinical diagnosis. This suggests that microbiome alterations may accumulate over time, potentially reflecting or contributing to neurodegenerative processes long before tremors or rigidity appear.
While the study does not establish causation, it supports the hypothesis that gut microbial changes could be part of a cascading biological pathway leading to alpha-synuclein misfolding, a hallmark of Parkinson’s pathology. Some researchers propose that misfolded proteins may originate in the gut and spread to the brain via the vagus nerve, a theory bolstered by epidemiological data showing lower Parkinson’s incidence in individuals who underwent early vagotomy.
Implications for Early Detection and Intervention
If validated in larger, longitudinal studies, microbiome signatures could one day complement genetic screening and imaging biomarkers in identifying individuals at highest risk. Unlike invasive procedures, stool sampling is non-invasive, repeatable, and relatively low-cost — making it attractive for population-level screening programs.
Experts caution, however, that microbiome composition is highly variable and influenced by diet, medications, geography, and lifestyle. Any future diagnostic application would likely require personalized baselines or machine learning models trained on diverse populations to distinguish meaningful signals from noise.
Nonetheless, the prospect of modulating the gut microbiome to delay or prevent disease onset is gaining traction. Interventions under investigation include targeted probiotics, prebiotic fibers, fecal microbiota transplantation, and dietary approaches like the Mediterranean diet, which has been linked to both improved gut health and lower neurodegenerative risk in observational studies.
As of 2024, clinical trials examining microbiome-based therapies for Parkinson’s are underway at institutions including Charité – Universitätsmedizin Berlin and the Mayo Clinic, though none have yet reported definitive results. The Michael J. Fox Foundation continues to support research in this area, emphasizing the need for rigorous, long-term validation before clinical adoption.
What This Means for Patients and Families
For individuals with a family history of Parkinson’s or known genetic risk factors, these findings underscore the importance of monitoring overall health — including gastrointestinal function — as part of a broader risk management strategy. Chronic constipation, for example, is one of the most common non-motor symptoms reported years before diagnosis and may reflect underlying gut microbiome alterations.
While no microbiome test is currently approved for predicting Parkinson’s disease, maintaining gut health through balanced nutrition, regular physical activity, and judicious antibiotic use remains a sensible, evidence-informed approach. Patients are encouraged to discuss any persistent digestive changes with their neurologist or primary care provider, especially when combined with other early signs like REM sleep behavior disorder or hyposmia (reduced sense of smell).
Advocacy groups such as the European Parkinson’s Disease Association stress that emerging science should be communicated with hope but without overpromise. “We’re seeing exciting connections between the gut and brain,” said a spokesperson for the organization in a 2023 public forum, “but we must avoid creating unnecessary anxiety. The goal is informed vigilance, not alarm.”
Looking Ahead: Research Priorities and Next Steps
The scientific community agrees that larger, longitudinal studies are essential to determine whether microbiome changes drive disease progression or are merely a byproduct. Ongoing initiatives like the Parkinson’s Progression Markers Initiative (PPMI), sponsored by the Michael J. Fox Foundation, are beginning to incorporate microbiome sampling into their protocols, which may yield clearer answers in the coming years.
Researchers also emphasize the need for greater diversity in study populations. Much of the current data comes from cohorts in North America and Europe, limiting understanding of how genetic, dietary, and environmental factors interact across global populations. Efforts to expand sampling in Asia, Africa, and Latin America are underway but remain underfunded relative to the disease burden.
For now, the evolving science of the microbiome in Parkinson’s disease serves as a reminder that neurological conditions may have origins far beyond the brain. As Dr. Helena Fischer notes, “When we look at the gut, we’re not just looking at digestion — we’re listening to a quiet conversation between microbes and the nervous system that may begin decades before a diagnosis is made.”
The next major update on microbiome biomarkers in Parkinson’s is expected from the International Congress of Parkinson’s Disease and Movement Disorders, scheduled for June 2027 in Kyoto, Japan. Until then, researchers and clinicians alike will continue to weigh the promise of microbial insights against the need for caution, rigor, and patient-centered communication.
We invite readers to share their thoughts or questions in the comments below. If you found this article informative, please consider sharing it with others who may benefit from understanding the latest developments in Parkinson’s research.