Switzerland’s federal government has released unredacted contracts for COVID-19 vaccines from Moderna and Novavax following a landmark court ruling, revealing the financial commitments made during the pandemic and reigniting public debate over vaccine procurement transparency.
The Federal Office of Public Health (BAG) published the full agreements in April 2026 after the Federal Administrative Court ruled in February that the contracts must be disclosed without redactions under Switzerland’s Freedom of Information Act. The decision became legally binding in early April, ending a prolonged legal challenge by transparency advocates who had sought access to the documents since late 2023.
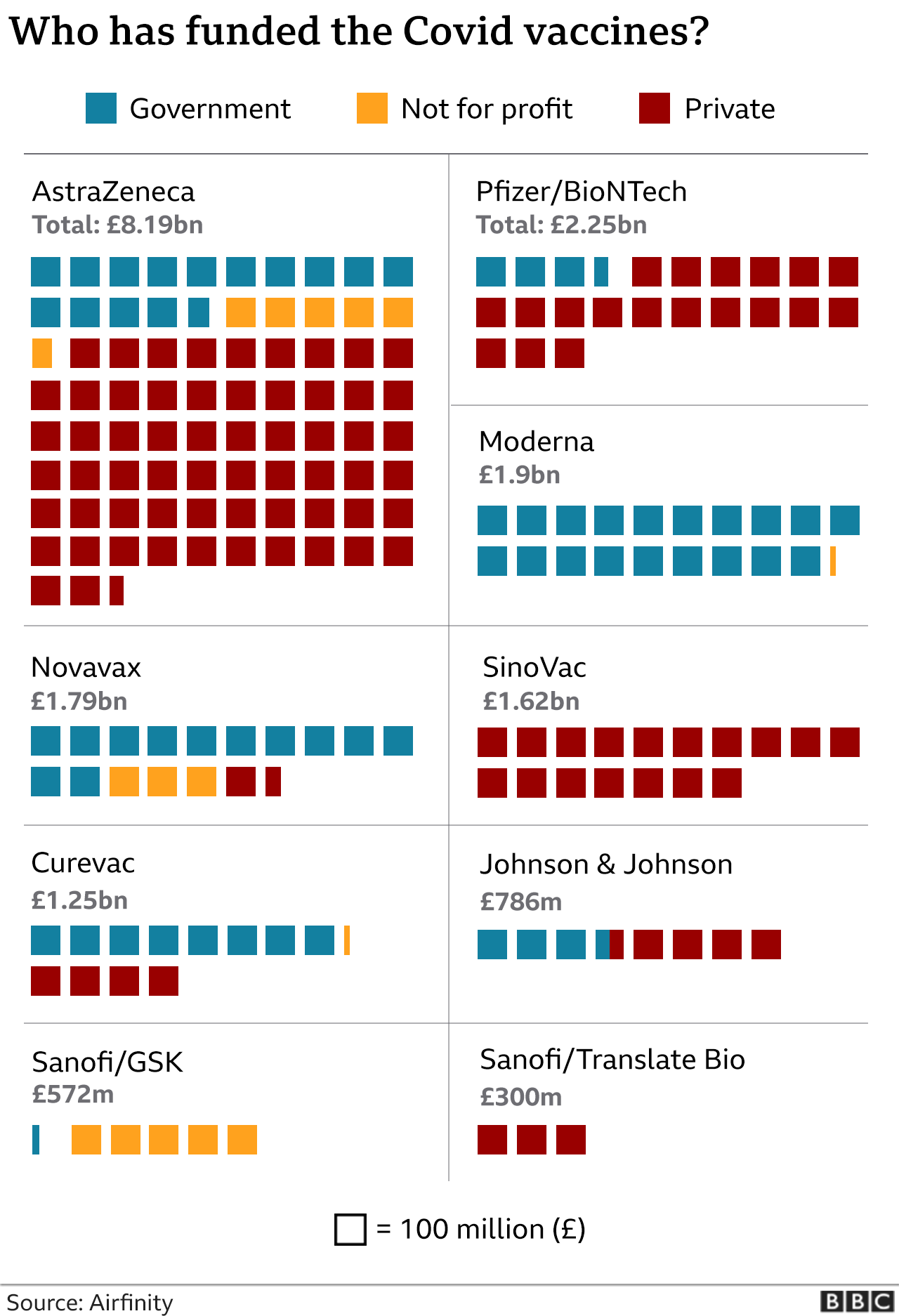
According to the published contracts, Switzerland secured 31 million doses of Moderna’s mRNA vaccine at a cost of approximately $32 per dose, resulting in a financial obligation of around $980 million. For Novavax’s protein-based vaccine, the government reserved one million doses at $22 per dose in December 2021, amounting to roughly $22 million. These figures were confirmed in BAG’s disclosures and reported by Swiss public broadcaster SRF.
The release of the contracts has drawn criticism from fiscal watchdogs and opposition politicians, who argue that the expenditures lacked sufficient parliamentary oversight and that the guarantees provided by manufacturers were unclear. The Solothurner Zeitung highlighted concerns over what it described as “billion-franc expenditures” without adequate accountability mechanisms in place during the emergency procurement phase.
BAG had previously released partially redacted versions of the contracts in 2022, citing fears that full disclosure could weaken Switzerland’s negotiating position in future pandemics. However, the court determined that the public’s right to access information under the Federal Act on the Principle of Government Transparency (BGÖ) outweighed those concerns, particularly given that the emergency phase of the pandemic had concluded.
The Moderna agreements include several key documents: a Memorandum of Understanding signed on June 9, 2020; the main Agreement dated August 5, 2020; a Parent Guarantee Agreement from the same date; a First Amendment from December 4, 2020; an Amended Parent Guarantee Agreement also from December 4, 2020; and a Second Amendment from February 2, 2021. All documents are in English and were published without redactions on the BAG website in April 2026.
Novavax’s contract, while less detailed in the public summaries, was also ordered to be fully disclosed. The agreement covered the procurement of a protein-subunit vaccine, which played a smaller role in Switzerland’s vaccination campaign compared to the mRNA options from Moderna and Pfizer-BioNTech.
Health policy experts note that the case underscores the tension between governmental transparency and the necessitate for agile decision-making during public health emergencies. While emergency procurement mechanisms allowed Switzerland to secure vaccines early, the lack of real-time oversight has prompted calls for reform in how such contracts are negotiated, approved, and archived.
The disclosed contracts are now permanently available on the BAG’s official website under the section dedicated to COVID-19 vaccine procurement. They remain the only coronavirus vaccine contracts fully published in unredacted form by the Swiss federal government to date, as agreements with other manufacturers such as Pfizer, Janssen, AstraZeneca, and CureVac were not subject to the same court order.
As of April 2026, no further legal challenges to the disclosure have been filed, and the BAG has not indicated plans to contest the ruling. The next procedural step involves the archiving of these documents as part of the federal record, with no scheduled hearings or updates anticipated unless recent freedom of information requests are submitted.
For readers seeking to review the primary sources, the Federal Office of Public Health provides direct access to all published contracts through its official portal. Journalists, researchers, and citizens are encouraged to consult these documents directly for the most accurate and complete information.
What do you think about the balance between emergency powers and public accountability in health crises? Share your perspective in the comments below, and facilitate spread informed discussion by sharing this article with others who follow global health policy developments.