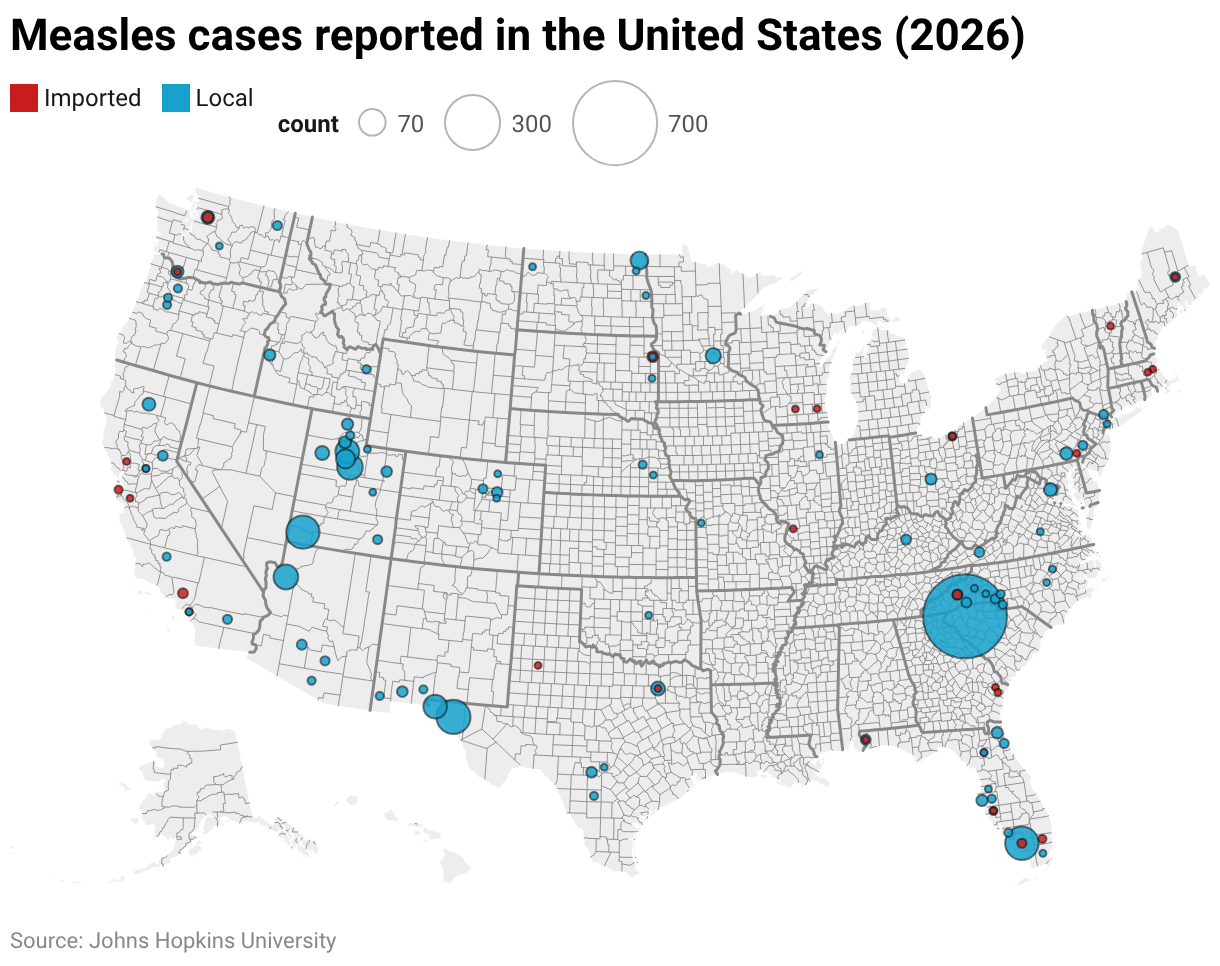
In March 2026, a federal judge issued a temporary injunction halting a directive from U.S. Health and Human Services Secretary Robert F. Kennedy Jr. To review the nation’s childhood immunization schedule, citing procedural concerns and potential risks to public health infrastructure. The ruling, delivered by U.S. District Judge Tanya Chutkan in the District of Columbia, prevents the HHS from implementing any changes to the Advisory Committee on Immunization Practices (ACIP)-recommended vaccine schedule even as litigation challenging the secretary’s authority proceeds. The decision marks a significant legal check on efforts to overhaul one of the country’s most enduring public health frameworks.
The injunction follows Kennedy’s December 2025 memo directing HHS staff to reevaluate the childhood vaccine schedule, a move that immediately drew criticism from medical associations, state health officials, and vaccine advocacy groups. Critics argued the directive bypassed established scientific review processes and risked undermining confidence in vaccines that have prevented millions of cases of measles, polio, and whooping cough. The judge’s ruling emphasized that any alteration to the immunization schedule must follow the Administrative Procedure Act, which requires notice, public comment, and evidence-based justification — steps the plaintiffs contended were sidestepped in the secretary’s initial order.
According to court filings reviewed by World Today Journal, the plaintiffs — including the American Academy of Pediatrics, the Association of State and Territorial Health Officials, and several parent advocacy coalitions — argued that Kennedy’s memo lacked transparency and violated federal law by attempting to override ACIP recommendations without convening the committee or publishing a formal proposal in the Federal Register. The judge agreed, stating in her 22-page order that “the Secretary’s directive appears to constitute a final agency action subject to judicial review, and the plaintiffs have shown a likelihood of success on the merits of their claim that the action was arbitrary, capricious, and an abuse of discretion.”
The childhood immunization schedule, updated annually by the CDC’s ACIP, serves as the foundation for school-entry requirements in all 50 states and the District of Columbia. It outlines the timing and dosage of vaccines for diseases such as hepatitis B, rotavirus, diphtheria, tetanus, pertussis, Haemophilus influenzae type b, pneumococcal conjugate, poliovirus, influenza, measles, mumps, rubella, varicella, and hepatitis A. The schedule also informs insurance coverage rules under the Affordable Care Act, which mandates that most private health plans cover ACIP-recommended vaccines without cost-sharing.
Legal experts note that the case hinges on whether the secretary possesses unilateral authority to alter a schedule developed through years of scientific consensus and peer-reviewed data. “The ACIP process is deliberately insulated from political influence to ensure vaccine recommendations are based solely on safety and efficacy,” said Dr. Lawrence Gostin, professor of global health law at Georgetown University. “Allowing a political appointee to override that process without transparency sets a dangerous precedent for public health decision-making.”
HHS has not yet announced whether it will appeal the injunction. In a brief statement to reporters, a department spokesperson said the agency is “reviewing the court’s decision and remains committed to ensuring vaccine policies are grounded in science and transparency.” The spokesperson did not confirm whether Kennedy intends to revise the directive or pursue alternative administrative pathways to achieve a review.
The ruling comes amid heightened scrutiny of federal health agencies following a series of leadership changes and policy shifts at HHS and the CDC since early 2025. While Kennedy has long expressed skepticism about certain vaccine components — a position he detailed during his 2024 presidential campaign and in subsequent public appearances — he has not publicly endorsed the removal of any currently recommended vaccine from the schedule. Instead, he has framed his review as an effort to examine “potential conflicts of interest” and “long-term safety data” associated with immunization programs.
Public health officials warn that even the perception of instability in vaccine policy could have tangible consequences. A 2025 study published in JAMA Network Open found that communities exposed to mixed messaging about vaccine safety experienced measles vaccination rate declines of up to 8 percentage points over 18 months, increasing outbreak risk. “Vaccine confidence is fragile,” said Dr. Saad Omer, director of the Yale Institute for Global Health. “When leaders signal doubt — even without changing policy — it can erode trust and deter parents from vaccinating their children on time.”
As the litigation continues, the status quo remains: the current childhood immunization schedule, last updated in February 2026, remains in effect for school enrollment and insurance coverage. Parents seeking official guidance are directed to the CDC’s immunization schedule page, which provides age-based vaccine recommendations and catch-up guidance for those who have fallen behind. State health departments continue to enforce school-entry requirements based on the ACIP schedule, with exemptions permitted only for medical or, in some states, non-medical reasons as defined by local law.
The next development in the case is expected in May 2026, when both parties are scheduled to submit preliminary injunction briefs ahead of a hearing set for June 10 before Judge Chutkan. Until then, the temporary restraining order remains in place, preserving the existing vaccine schedule while the courts determine the limits of executive authority over public health science.
For readers seeking to stay informed, the World Today Journal Health section will continue to monitor developments in this case and provide updates as new filings emerge or rulings are issued. We encourage you to share this article with others who may be affected by changes to vaccine policy and to consult your healthcare provider or local public health department for personalized immunization advice.
In March 2026, a federal judge issued a temporary injunction halting a directive from U.S. Health and Human Services Secretary Robert F. Kennedy Jr. To review the nation’s childhood immunization schedule, citing procedural concerns and potential risks to public health infrastructure. The ruling, delivered by U.S. District Judge Tanya Chutkan in the District of Columbia, prevents the HHS from implementing any changes to the Advisory Committee on Immunization Practices (ACIP)-recommended vaccine schedule while litigation challenging the secretary’s authority proceeds. The decision marks a significant legal check on efforts to overhaul one of the country’s most enduring public health frameworks.
The injunction follows Kennedy’s December 2025 memo directing HHS staff to reevaluate the childhood vaccine schedule, a move that immediately drew criticism from medical associations, state health officials, and vaccine advocacy groups. Critics argued the directive bypassed established scientific review processes and risked undermining confidence in vaccines that have prevented millions of cases of measles, polio, and whooping cough. The judge’s ruling emphasized that any alteration to the immunization schedule must follow the Administrative Procedure Act, which requires notice, public comment, and evidence-based justification — steps the plaintiffs contended were sidestepped in the secretary’s initial order.
According to court filings reviewed by World Today Journal, the plaintiffs — including the American Academy of Pediatrics, the Association of State and Territorial Health Officials, and several parent advocacy coalitions — argued that Kennedy’s memo lacked transparency and violated federal law by attempting to override ACIP recommendations without convening the committee or publishing a formal proposal in the Federal Register. The judge agreed, stating in her 22-page order that “the Secretary’s directive appears to constitute a final agency action subject to judicial review, and the plaintiffs have shown a likelihood of success on the merits of their claim that the action was arbitrary, capricious, and an abuse of discretion.”
The childhood immunization schedule, updated annually by the CDC’s ACIP, serves as the foundation for school-entry requirements in all 50 states and the District of Columbia. It outlines the timing and dosage of vaccines for diseases such as hepatitis B, rotavirus, diphtheria, tetanus, pertussis, Haemophilus influenzae type b, pneumococcal conjugate, poliovirus, influenza, measles, mumps, rubella, varicella, and hepatitis A. The schedule also informs insurance coverage rules under the Affordable Care Act, which mandates that most private health plans cover ACIP-recommended vaccines without cost-sharing.
Legal experts note that the case hinges on whether the secretary possesses unilateral authority to alter a schedule developed through years of scientific consensus and peer-reviewed data. “The ACIP process is deliberately insulated from political influence to ensure vaccine recommendations are based solely on safety and efficacy,” said Dr. Lawrence Gostin, professor of global health law at Georgetown University. “Allowing a political appointee to override that process without transparency sets a dangerous precedent for public health decision-making.”
HHS has not yet announced whether it will appeal the injunction. In a brief statement to reporters, a department spokesperson said the agency is “reviewing the court’s decision and remains committed to ensuring vaccine policies are grounded in science and transparency.” The spokesperson did not confirm whether Kennedy intends to revise the directive or pursue alternative administrative pathways to achieve a review.
The ruling comes amid heightened scrutiny of federal health agencies following a series of leadership changes and policy shifts at HHS and the CDC since early 2025. While Kennedy has long expressed skepticism about certain vaccine components — a position he detailed during his 2024 presidential campaign and in subsequent public appearances — he has not publicly endorsed the removal of any currently recommended vaccine from the schedule. Instead, he has framed his review as an effort to examine “potential conflicts of interest” and “long-term safety data” associated with immunization programs.
Public health officials warn that even the perception of instability in vaccine policy could have tangible consequences. A 2025 study published in JAMA Network Open found that communities exposed to mixed messaging about vaccine safety experienced measles vaccination rate declines of up to 8 percentage points over 18 months, increasing outbreak risk. “Vaccine confidence is fragile,” said Dr. Saad Omer, director of the Yale Institute for Global Health. “When leaders signal doubt — even without changing policy — it can erode trust and deter parents from vaccinating their children on time.”
As the litigation continues, the status quo remains: the current childhood immunization schedule, last updated in February 2026, remains in effect for school enrollment and insurance coverage. Parents seeking official guidance are directed to the CDC’s immunization schedule page, which provides age-based vaccine recommendations and catch-up guidance for those who have fallen behind. State health departments continue to enforce school-entry requirements based on the ACIP schedule, with exemptions permitted only for medical or, in some states, non-medical reasons as defined by local law.
The next development in the case is expected in May 2026, when both parties are scheduled to submit preliminary injunction briefs ahead of a hearing set for June 10 before Judge Chutkan. Until then, the temporary restraining order remains in place, preserving the existing vaccine schedule while the courts determine the limits of executive authority over public health science.
For readers seeking to stay informed, the World Today Journal Health section will continue to monitor developments in this case and provide updates as new filings emerge or rulings are issued. We encourage you to share this article with others who may be affected by changes to vaccine policy and to consult your healthcare provider or local public health department for personalized immunization advice.