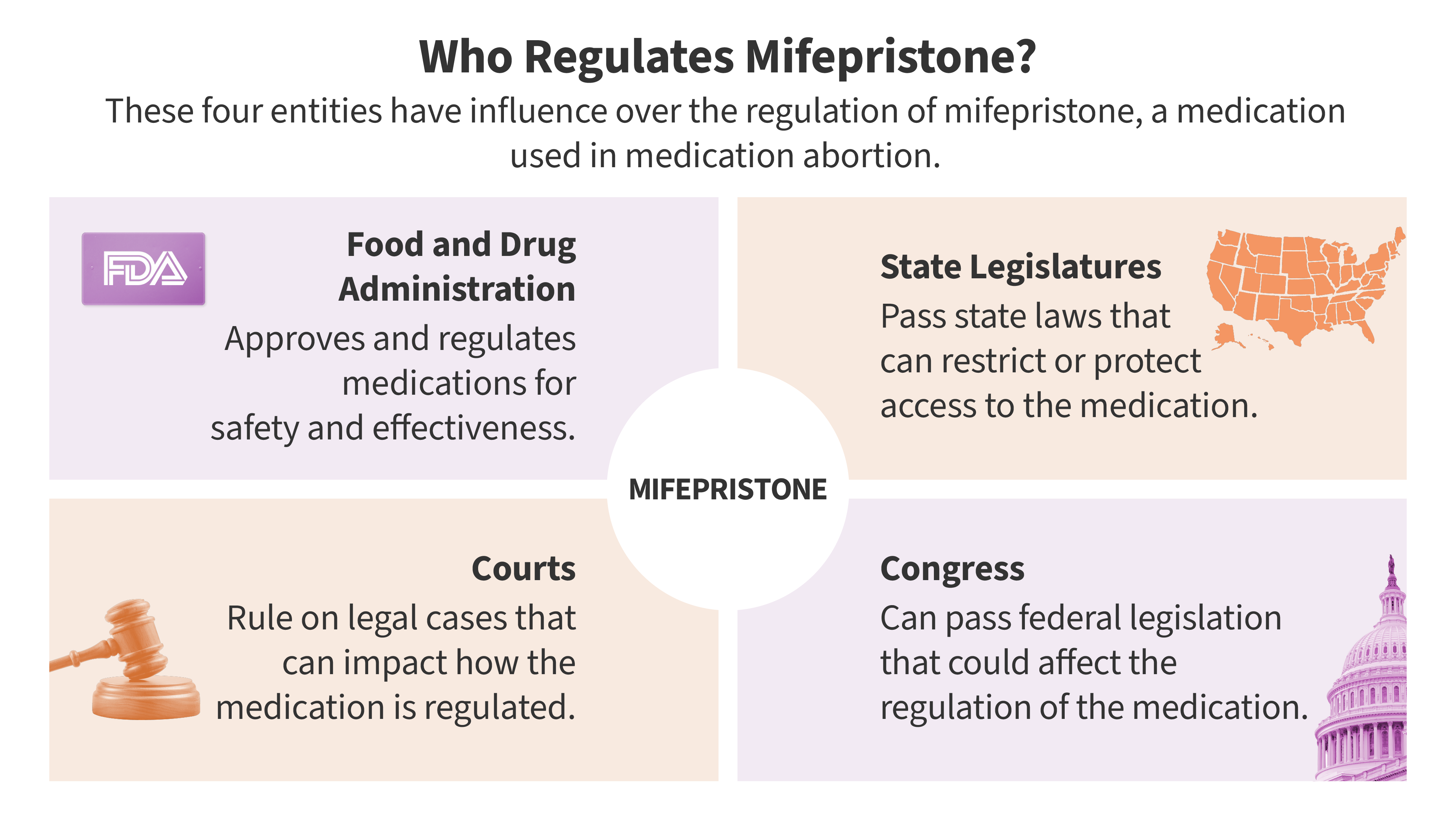
The intersection of federal drug regulation and state-level abortion bans has created one of the most complex legal landscapes in modern American medicine. At the center of this conflict is mifepristone, a medication that has become the primary vehicle for medication abortions in the United States. The legal battle, which reached the U.S. Supreme Court through various challenges including those involving the state of Louisiana, represents more than just a dispute over a pill. it is a fundamental clash over who controls the administration of approved pharmaceuticals.
For healthcare providers and patients, the uncertainty surrounding mifepristone access has created a fragmented system of care. In states where abortion is banned or severely restricted, the ability to obtain this medication via telehealth or mail has often been the only viable option for early pregnancy termination. The legal challenges brought by Louisiana and other anti-abortion advocates sought to roll back the U.S. Food and Drug Administration (FDA) regulations that expanded this access, arguing that the drug’s distribution posed undue safety risks.
As a physician and health journalist, I have watched this case unfold not only as a legal drama but as a public health crisis. When the availability of a clinically safe and FDA-approved medication is jeopardized by litigation, the result is often an increase in unregulated alternatives or delayed care, both of which compromise patient safety. The resolution of these challenges provides a critical window into how federal authority over drug safety interacts with the current state-led restrictions on reproductive healthcare.
The core of the dispute centers on the FDA’s decision to ease restrictions on mifepristone over the last decade. Originally approved in 2000, the drug was initially subject to strict requirements, including in-person dispensing by a certified provider. However, in 2016 and again in 2021, the FDA updated its regulations to allow the drug to be prescribed via telehealth and sent through the mail, reflecting evolving medical standards and the need for broader patient access.
The Medical Role of Mifepristone in Abortion Care
To understand the legal stakes, it is first necessary to understand what mifepristone is from a clinical perspective. Mifepristone is a progesterone receptor antagonist. In a pregnancy, progesterone is the hormone required to maintain the lining of the uterus; by blocking this hormone, mifepristone causes the lining to break down, effectively ending the pregnancy’s viability. It is typically followed 24 to 48 hours later by misoprostol, a prostaglandin that causes the uterus to contract and expel the pregnancy tissue.
From a public health standpoint, this medication abortion regimen is highly effective and has a strong safety profile when used according to FDA guidelines. According to the U.S. Food and Drug Administration, the drug is approved for use up to 70 days (10 weeks) of gestation. The shift toward telehealth and mail-order delivery was not a sudden political move, but a response to the medical community’s consensus that the drug can be safely managed remotely with proper screening and follow-up.
The challenge brought by Louisiana and other plaintiffs argued that the FDA ignored safety concerns—specifically regarding the risk of hemorrhage or incomplete abortion—by allowing the drug to be prescribed without an in-person exam. However, major medical organizations, including the American College of Obstetricians and Gynecologists (ACOG), have consistently maintained that telehealth is a safe and effective method for delivering medication abortion.
The Legal Journey to the Supreme Court
The litigation involving mifepristone followed a turbulent path through the federal court system. The primary challenge was spearheaded by the Alliance for Hippocratic Medicine, a group of anti-abortion physicians, and supported by several states, including Louisiana. They sought to have the FDA reverse its 2000 approval of the drug or, at the very least, reinstate the restrictive dispensing requirements of the early 2000s.
The case moved rapidly through the courts, eventually reaching the U.S. Supreme Court in the case known as FDA v. Alliance for Hippocratic Medicine. The legal strategy of the plaintiffs relied on the claim that the FDA’s actions were “arbitrary and capricious” under the Administrative Procedure Act, suggesting that the agency had failed to properly consider the risks of the drug when it expanded access.

In a pivotal ruling on June 13, 2024, the Supreme Court of the United States ruled unanimously that the plaintiffs lacked the legal standing to bring the lawsuit. “Standing” is a fundamental legal requirement; it means the party bringing the suit must have suffered a direct, concrete injury. The Court found that the physicians and the supporting states did not demonstrate that they were personally harmed by the FDA’s regulation of the drug.
This ruling was a significant victory for reproductive rights advocates and the FDA. By dismissing the case on standing, the Court avoided a ruling on the merits of the drug’s safety or the FDA’s authority, but the practical effect was immediate: the current FDA regulations allowing for telehealth and mail-order mifepristone remained in place.
The Clash Between State Laws and Federal Regulation
While the Supreme Court’s ruling on standing preserved the FDA’s rules, it did not resolve the underlying tension between state and federal law. This is where the role of states like Louisiana becomes particularly complex. Many states have passed laws that ban abortion entirely or restrict it to very narrow circumstances. These state laws often conflict with the federal government’s approval of a medication designed specifically for that purpose.
This creates a “legal gray zone” for both providers and patients. While the FDA says mifepristone can be mailed, a state law may criminalize the act of providing that medication. This has led to the rise of “shield laws” in some states, which protect providers who mail medication to patients in states where abortion is illegal. Conversely, states like Louisiana have sought to use the court system to force the federal government to restrict the drug, effectively attempting to use federal regulation to enforce state-level bans.
For the global community, this situation highlights a unique American phenomenon: the fragmentation of medical standards based on geography. In most developed nations, the approval of a medication by a national health authority ensures uniform access and standards of care. In the U.S., the Dobbs v. Jackson decision, which overturned Roe v. Wade, shifted the authority to regulate abortion to the states, creating a scenario where a federally approved drug is legal in one zip code and a felony in another.
What This Means for Patient Access and Safety
The immediate impact of the Supreme Court’s decision is the preservation of the status quo. Patients who rely on telehealth and mail-order mifepristone can continue to access the medication through providers who operate under the FDA’s current guidelines. This is particularly critical for those in “abortion deserts”—areas where the nearest clinic is hundreds of miles away.
However, the legal instability continues to have a chilling effect on medical practice. Many physicians remain hesitant to prescribe mifepristone for fear of prosecution under state laws, even if they are following federal guidelines. This hesitation can lead to delays in care, which is a clinical concern; medication abortion is most effective and safest when administered early in the first trimester.
the ongoing legal volatility encourages some patients to seek mifepristone from unverified online sources. As a physician, this is my greatest concern. While mifepristone itself is safe, medications obtained from unregulated sources may be counterfeit, contaminated, or incorrectly dosed. The most effective way to ensure patient safety is to maintain clear, legal, and accessible pathways to licensed healthcare providers.
Key Takeaways on the Mifepristone Legal Battle
- The Ruling: The Supreme Court ruled in June 2024 that the plaintiffs in the mifepristone case lacked standing, meaning the FDA’s current access rules remain active.
- FDA Regulations: Current rules allow mifepristone to be prescribed via telehealth and delivered by mail, removing the requirement for an in-person clinic visit.
- Clinical Safety: Mifepristone is an FDA-approved medication with a high safety profile, acting as a progesterone blocker to terminate pregnancy.
- State vs. Federal: A fundamental conflict persists between federal drug approval (FDA) and state-level abortion bans (e.g., in Louisiana).
- Patient Impact: The ruling prevents an immediate nationwide restriction of the drug, but state-level criminalization still poses risks to providers and patients.
Future Outlook and Next Steps
While the June 2024 ruling ended this specific legal challenge, the battle over mifepristone is unlikely to be over. Anti-abortion advocates may attempt to bring new lawsuits with different plaintiffs who can demonstrate “standing,” or they may continue to push for state-level legislation that targets the distribution of the drug within their borders.
The medical community will continue to monitor the impact of these legal shifts on maternal health outcomes. The focus remains on ensuring that evidence-based medicine prevails over political litigation. For patients seeking official information, the FDA’s official website remains the authoritative source for medication guidelines and safety warnings.
The next critical checkpoints will be the emergence of any new challenges to the FDA’s authority in lower federal courts and the potential for further state-level “shield laws” to expand. As the legal landscape evolves, the priority must remain the safety and autonomy of the patient.
Do you believe federal drug approvals should override state restrictions on medication? Share your thoughts in the comments below or share this article to keep others informed on the current state of reproductive healthcare access.