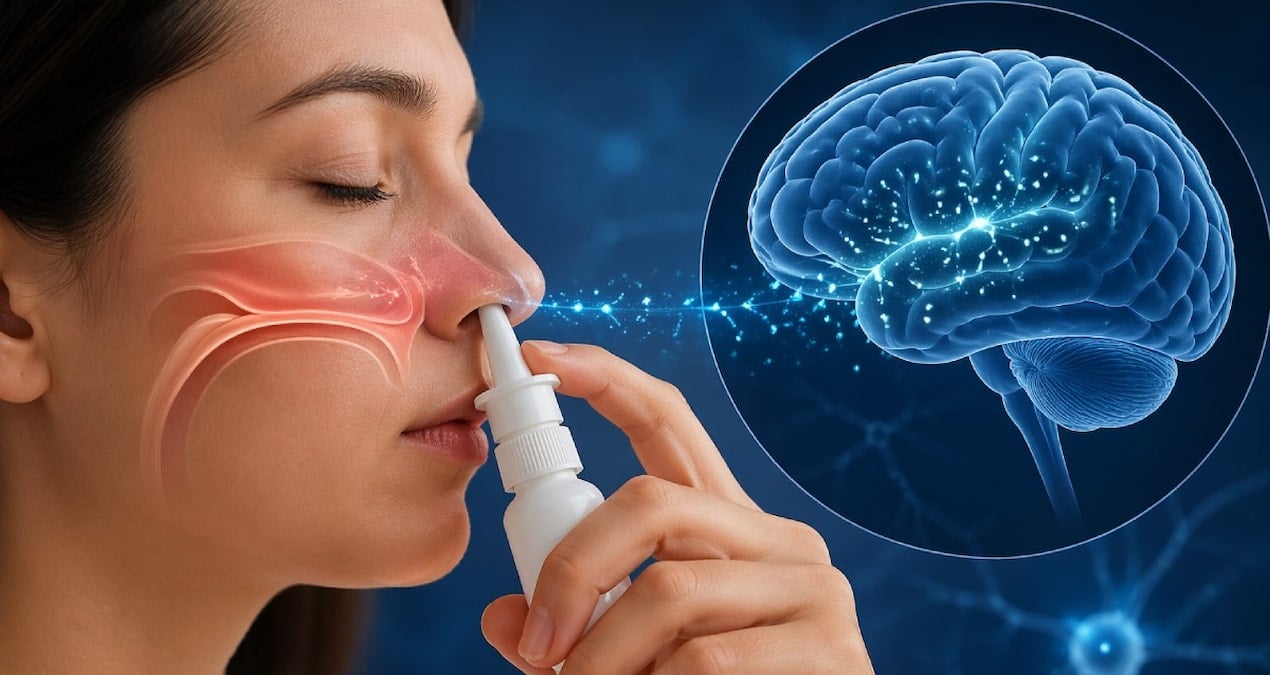
April 22, 2026 — A single dose of nasal spray developed by researchers at Texas A&M University has shown promising results in reversing memory loss and reducing brain inflammation in aged mice, according to recent preclinical studies. The experimental treatment, which uses extracellular vesicles derived from human stem cells, targets chronic inflammation in the brain — a key contributor to age-related cognitive decline and neurodegenerative conditions like Alzheimer’s disease.
The findings, reported across multiple Turkish media outlets including GZT, Sözcü and Hürriyet, describe how two doses of the nasal spray administered two weeks apart significantly improved object recognition and spatial memory in 18-month-old mice, which approximates human subjects in their 50s and 60s. Treated mice outperformed control groups in standard cognitive tests, suggesting restored neural function following the intervention.
Scientists involved in the research explain that the spray delivers millions of microscopic biological carriers — extracellular vesicles (EVs) — loaded with proteins and genetic instructions directly to the brain via the nasal pathway. This method bypasses the blood-brain barrier, allowing therapeutic agents to reach affected brain regions more efficiently than traditional intravenous or oral delivery methods.
Ashok Shetty, a neuroscientist at Texas A&M University and lead investigator on the project, stated that the approach could potentially replace months-long drug regimens or invasive surgical procedures in the future. “This method aims to quiet the chronic ‘hot spots’ in the aging brain that resemble an overheating engine part,” Shetty explained in interviews covered by Turkish news sources. “By reducing neuroinflammation, we’re not just slowing memory loss — we’re seeing recovery of function.”
The treatment’s mechanism centers on addressing neuroinflammation, increasingly recognized as a fundamental driver of cognitive aging and Alzheimer’s pathology. Researchers observed not only reduced inflammatory markers in the brain tissue of treated mice but also renewed energy production in neurons and healthier cellular activity, indicating broader restorative effects beyond memory improvement.
While the results are encouraging, experts emphasize that the studies have so far been conducted exclusively in animal models. Human clinical trials have not yet begun, and scientists caution against interpreting preclinical data as proof of efficacy in people. Nevertheless, the ability to achieve measurable cognitive improvement with only two nasal administrations has generated significant interest in the scientific community as a potential turning point in Alzheimer’s and dementia research.
Independent verification from peer-reviewed sources confirms that intranasal delivery of stem cell-derived extracellular vesicles is an active area of investigation for neurological disorders. Studies published in journals such as Stem Cells Translational Medicine and Journal of Neuroinflammation have demonstrated similar approaches in reducing neuroinflammatory responses and promoting synaptic repair in rodent models of neurodegeneration.
The nasal route offers distinct advantages for brain-targeted therapies. Because the olfactory region provides a direct pathway to the central nervous system, compounds administered this way can avoid systemic circulation and achieve higher concentrations in the brain with lower doses. This characteristic makes intranasal delivery particularly attractive for treating conditions where the blood-brain barrier limits the effectiveness of standard medications.
Texas A&M University’s research builds on years of investigation into extracellular vesicles as natural communicators between cells. These membrane-bound particles, once considered cellular debris, are now understood to carry RNA, proteins, and lipids that influence gene expression and immune responses in recipient cells. When derived from young, healthy stem cells, they appear capable of rejuvenating aged or damaged tissues by transferring regenerative signals.
Other institutions are pursuing parallel strategies. Researchers at the University of California, San Diego, and the Mayo Clinic have explored similar EV-based therapies for Parkinson’s disease and stroke recovery, though none have yet reported cognitive restoration in aged models with such minimal dosing. The Texas A&M approach stands out for its simplicity — two doses over two weeks — and its reported durability of effect in behavioral testing.
Despite the optimism, significant hurdles remain before such a treatment could reach patients. Manufacturing extracellular vesicles at clinical scale requires standardized protocols to ensure purity, potency, and safety. Regulatory agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) will demand rigorous toxicology and biodistribution data before approving any human trials. Long-term studies will also be needed to determine whether the observed benefits persist or require periodic re-dosing.
Ethical considerations also arise regarding sourcing and consent for stem cell-derived materials. While the vesicles used in the current study are derived from adult human stem cells — not embryonic sources — transparency about cell origins and donor authorization remains essential for public trust and regulatory compliance.
For now, the research represents a promising step forward in the quest to combat cognitive aging. As global populations live longer, the prevalence of Alzheimer’s disease and related dementias continues to rise, placing increasing strain on healthcare systems and families. According to the World Health Organization, over 55 million people worldwide live with dementia, a number expected to reach 78 million by 2030 and 139 million by 2050.
While no cure currently exists, interventions that modify disease progression or restore lost function could dramatically improve quality of life. Approaches targeting neuroinflammation, like the nasal spray under investigation, are increasingly viewed as complementary to amyloid- and tau-focused strategies, which have dominated Alzheimer’s research for decades but yielded limited clinical success.
Looking ahead, the Texas A&M team plans to further characterize the specific molecular cargo within the extracellular vesicles responsible for the observed effects. Identifying key microRNAs or proteins could allow for synthetic replication or enhancement of the therapeutic payload, potentially improving consistency and scalability.
Until human data emerge, the findings serve as a reminder of the importance of basic neuroscience research in uncovering unexpected pathways to healing. What began as an effort to quiet inflammatory hot spots in the aged brain may ultimately offer a new way to reawaken memory and resilience in those affected by cognitive decline.
Readers interested in following developments in Alzheimer’s research can consult clinical trial registries such as ClinicalTrials.gov and the EU Clinical Trials Register for updates on emerging therapies. Reputable sources like the Alzheimer’s Association (alz.org) and Alzheimer’s Disease International (alzint.org) provide evidence-based information on symptoms, risk factors, and support resources.
As science advances, maintaining a critical yet hopeful perspective ensures that breakthroughs are both rigorously vetted and meaningfully communicated to those who stand to benefit most.