Berlin, Germany — May 14, 2026 — A once-daily oral pill could soon replace injections for millions of people struggling to maintain weight loss after stopping GLP-1 therapies like Wegovy or Zepbound. The phase 3b ATTAIN-MAINTAIN trial, presented at the European Congress on Obesity, demonstrates that orforglipron—marketed as Foundayo—significantly preserves weight reduction and cardiometabolic benefits when transitioning from injectable GLP-1 receptor agonists to an oral maintenance strategy.
For the first time, researchers have shown that switching from injectable weight-loss drugs to an oral alternative can sustain up to 79% of initial weight loss over 52 weeks—a stark contrast to the 38% retention rate observed with placebo. The findings, published in a peer-reviewed medical journal, mark a pivotal moment in obesity treatment, potentially expanding access to GLP-1 therapies for patients who face barriers to injectable medications.
Dr. Helena Fischer, Editor of Health at World Today Journal, notes: “This trial addresses a critical gap in obesity management. While injectable GLP-1 drugs like semaglutide and tirzepatide have revolutionized weight loss, many patients struggle with adherence once they stop injections. Orforglipron offers a practical, scalable solution—especially in regions where injection infrastructure is limited.”
How Orforglipron Works: Bridging the Maintenance Gap
Orforglipron, developed by Eli Lilly and Company in collaboration with Chugai Pharmaceutical, is the first nonpeptide, small-molecule GLP-1 receptor agonist approved for oral administration. Unlike injectable GLP-1 drugs, which require weekly or daily injections, orforglipron is taken as a single daily pill, improving patient adherence and reducing injection-related side effects like pain or discomfort.
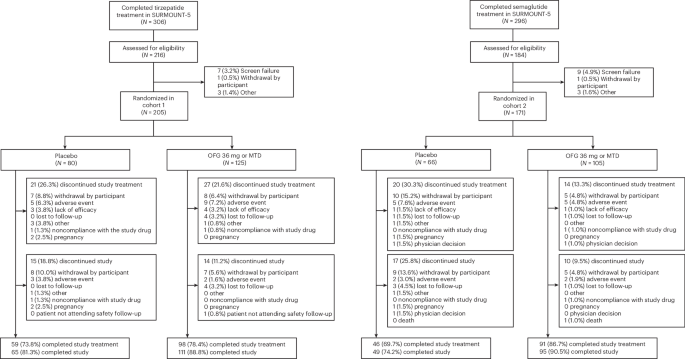
The ATTAIN-MAINTAIN trial enrolled 376 participants who had previously achieved weight loss with either tirzepatide (Zepbound) (Cohort 1: 205 patients) or semaglutide (Wegovy) (Cohort 2: 171 patients). After reaching a weight plateau, participants were randomized to receive either orforglipron 60mg once daily or a placebo for 52 weeks.
Key Findings: Weight Loss Retention and Cardiometabolic Benefits
The results are striking:
- Cohort 1 (tirzepatide → orforglipron):
- Participants maintained 74.7% (±4.05%) of their initial weight loss with orforglipron vs. 49.2% (±3.92%) with placebo.
- Treatment difference: 25.5 percentage points (95% CI: 14.5–36.5; P < 0.001).
- Cohort 2 (semaglutide → orforglipron):
- Participants maintained 79.3% (±4.42%) of their initial weight loss with orforglipron vs. 37.6% (±7.46%) with placebo.
- Treatment difference: 41.7 percentage points (95% CI: 24.4–59.0; P < 0.001).
Beyond weight retention, orforglipron also demonstrated improvements in:
- Blood pressure (systolic/diastolic reductions of ~5–7 mmHg vs. Placebo).
- Fasting glucose levels (reductions of ~10–15 mg/dL).
- Lipid profiles (reductions in LDL cholesterol and triglycerides).
Note: The trial did not include a comparator arm for continued injectable therapy, so direct comparisons between orforglipron and ongoing injections cannot be made from these data.
Why This Matters: Scalability and Patient Access
GLP-1 receptor agonists have transformed obesity treatment, but their widespread adoption faces challenges:
- Injection fatigue: Up to 30% of patients discontinue injectable therapies due to inconvenience or side effects like nausea or injection-site reactions (source).
- Global disparities: In low- and middle-income countries, injection infrastructure (e.g., refrigeration, trained healthcare providers) limits access to injectable drugs.
- Cost and adherence: Oral alternatives may improve long-term adherence, reducing healthcare costs associated with weight regain.
Orforglipron’s approval in the U.S. In April 2026 (under the brand name Foundayo) follows a decade of research into oral GLP-1 agonists. The drug’s mechanism—mimicking the natural hormone GLP-1 to regulate appetite and insulin secretion—mirrors that of injectable drugs but with the convenience of a pill.
Safety Profile: Gastrointestinal Effects as the Primary Concern
The most common adverse events with orforglipron were gastrointestinal in nature, including:
- Nausea (reported in ~25% of patients, mostly mild to moderate).
- Diarrhea or constipation (reported in ~15% of patients).
- No significant increases in pancreatitis, gallbladder disease, or thyroid C-cell tumors were observed in the 52-week trial.
These side effects align with the known safety profile of GLP-1 receptor agonists and were generally manageable, with most events resolving within weeks of treatment initiation.
What Happens Next: Regulatory and Real-World Adoption
With the ATTAIN-MAINTAIN trial results now published, several key developments are expected:
- EU and global regulatory submissions: Lilly has begun rolling submissions to the European Medicines Agency (EMA) and other regulators for orforglipron’s approval in obesity management. A decision is anticipated by late 2026 or early 2027.
- Real-world evidence studies: Observational data will be critical to assess orforglipron’s long-term efficacy (>2 years) and comparative effectiveness against continued injectable therapy.
- Insurance coverage and pricing: U.S. Insurers are likely to classify orforglipron as a maintenance therapy, potentially reducing out-of-pocket costs for patients compared to injectable drugs.
Who Benefits Most?
Orforglipron is poised to benefit several patient groups:
- Patients who achieved weight loss with injectables but struggle with adherence.
- Individuals in regions with limited injection infrastructure.
- Those with injection phobia or needle-related anxiety.
- Patients with cardiometabolic conditions (e.g., type 2 diabetes, hypertension) who need long-term GLP-1 therapy.
Exclusion: The trial did not enroll patients with a history of pancreatitis or medullary thyroid carcinoma, so safety in these populations remains unproven.
Expert Perspective: The Future of Oral GLP-1 Therapies
Dr. Sarah Kaplan, an endocrinologist at Massachusetts General Hospital, calls the results “a game-changer for maintenance therapy.” She notes: “The ability to switch from an injection to an oral pill without significant weight regain is a major advancement. For many patients, this could mean the difference between sustained health benefits and relapse into obesity-related complications.”
However, Dr. Kaplan cautions that orforglipron is not a “magic bullet.” “Patients will still need to combine this medication with lifestyle changes—diet and exercise—to maximize long-term success,” she says.
Key Takeaways
- Orforglipron preserves 74–79% of initial weight loss over 52 weeks in patients transitioning from injectable GLP-1 therapies.
- It offers a scalable, oral alternative to injections, addressing adherence and access barriers.
- Common side effects are gastrointestinal (nausea, diarrhea), typically mild to moderate.
- Regulatory approval in the EU and other regions is expected in 2027, pending submissions.
- Long-term data (>2 years) and real-world studies will be critical to confirm durability.
Where to Find Official Updates
For the latest information on orforglipron (Foundayo), patients and clinicians can consult:

- Lilly’s official Foundayo website (product details, clinical trials, and patient resources).
- ClinicalTrials.gov (trial protocol and results).
- European Medicines Agency (EMA) (for EU regulatory updates).
- U.S. FDA (post-approval safety monitoring).
Reader Questions: What You Need to Know
Q: Can orforglipron replace injectable GLP-1 drugs entirely?
A: The ATTAIN-MAINTAIN trial only tested orforglipron as a maintenance therapy after patients had already lost weight with injectables. Whether it can initiate weight loss (like semaglutide or tirzepatide) is not yet known and will require further trials.
Q: How much does orforglipron cost, and will insurance cover it?
A: In the U.S., Foundayo is priced at approximately $300–$400 per month (similar to other GLP-1 drugs). Coverage will depend on insurance plans, but many U.S. Insurers classify it as a weight management medication, often covering part or all of the cost for eligible patients.
Q: Are there other oral GLP-1 drugs in development?
A: Yes. Other oral GLP-1 agonists in late-stage trials include:
- Retatrutide (Harmony Biosciences) (a triple agonist targeting GLP-1, GIP, and glucagon receptors).
- Cagrilintide (BMS) (a dual GLP-1/amylin agonist).
- Ozempic Oral (semaglutide oral formulation, Novo Nordisk) (currently in phase 3 trials).
Next Steps: What to Watch For
The obesity treatment landscape is evolving rapidly. Key milestones to monitor include:
- June–December 2026: EMA and other global regulators review orforglipron’s approval dossier.
- 2027: Potential launch in the EU and other markets, with pricing and reimbursement decisions.
- Ongoing: Real-world data on orforglipron’s effectiveness in diverse populations (e.g., older adults, those with severe obesity).
For now, patients considering orforglipron should consult their healthcare provider to discuss whether it’s an appropriate option for their weight management journey.
Your Thoughts: Share Your Experience
Have you or a loved one used injectable GLP-1 drugs for weight loss? What challenges have you faced with adherence? Share your story in the comments below—or tag @WorldTodayJrnl on X/Twitter with your experiences.
Like this article? Share it with someone who could benefit from these findings. For more on medical innovation and public health, subscribe to World Today Journal’s Health newsletter.