For individuals living with advanced retinitis pigmentosa, the world often fades into a narrow tunnel of vision before slipping into profound darkness. This inherited retinal disease systematically destroys the light-sensing cells of the eye, leaving patients with limited to no functional sight. However, a recent first-in-human clinical trial suggests a potential recent path forward through intravitreal photoswitch therapy in advanced retinitis pigmentosa, aiming to bypass damaged cells and restore light responsiveness.
The study, known as the ABACUS-1 trial, evaluated a modest molecule called KIO-301. Unlike traditional gene therapies that attempt to fix a specific genetic mutation, this “photoswitch” approach is gene-agnostic, meaning it is designed to work regardless of the underlying genetic cause of the disease. By targeting the retinal ganglion cells—which normally sit behind the primary photoreceptors—the therapy seeks to turn these remaining cells into new, light-sensitive receptors published in Nature Medicine.
The results of this Phase 1 trial indicate that the therapy can be administered safely and is well-tolerated, providing a critical foundation for larger studies. While the trial was primarily focused on safety, exploratory data revealed signals that the treatment may indeed be influencing how the brain perceives light in patients who were previously considered to have late-stage vision loss.
Understanding the Photoswitch Mechanism
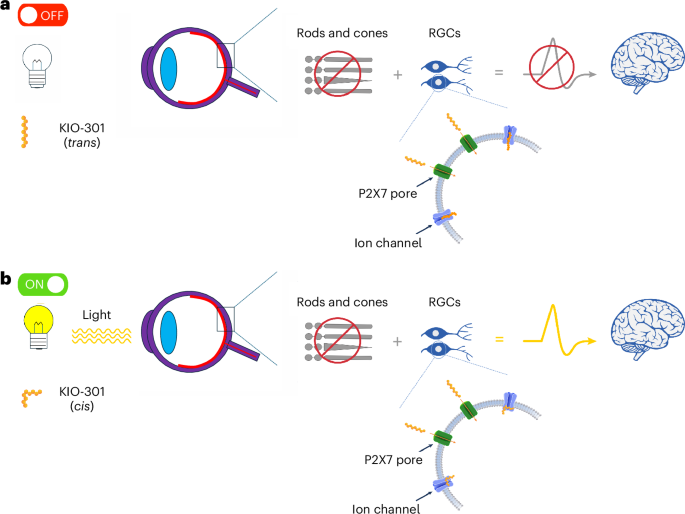
To understand why this therapy is significant, it is necessary to gaze at how retinitis pigmentosa affects the eye. In a healthy eye, rods and cones capture light and pass that signal to retinal ganglion cells, which then send the information to the brain’s visual cortex. In advanced retinitis pigmentosa, the rods and cones are lost, breaking the chain of communication.
KIO-301 is an azobenzene photoswitch molecule. When injected directly into the vitreous of the eye, it is designed to confer light responsiveness directly to the retinal ganglion cells. Essentially, the molecule acts as a surrogate photoreceptor, allowing the eye to capture light and transmit a signal to the brain even when the primary light-sensing cells are gone. This approach represents a shift in medical innovation, moving from repairing damaged genetics to augmenting the existing cellular architecture of the retina.
The ABACUS-1 Trial: Design and Safety Outcomes
The ABACUS-1 study was an open-label, dose-escalation trial designed to test the feasibility of this molecular approach. The trial involved 6 participants, with the therapy administered to a total of 12 eyes via intravitreal injection. Because the therapy is gene-agnostic, it was tested in individuals with late-stage disease irrespective of their specific genetic mutation.
The primary objective was to monitor ocular and systemic safety over a 30-day period. According to the trial data, the primary safety outcome was met, with no serious adverse events or dose-limiting toxicities observed at any point during the study. The researchers reported that no drug-related intraocular inflammation occurred, and all observed ocular adverse events were classified as mild and related to the injection procedure itself as detailed in the study results.
Key Clinical Observations
- Participant Cohort: 6 individuals with advanced retinitis pigmentosa (12 eyes total).
- Safety Window: Primary monitoring focused on the first 30 days post-injection.
- Tolerability: No serious adverse events or drug-induced inflammation reported.
- Administration: Delivered via intravitreal injection.
Exploratory Signals and Brain Activity
While Phase 1 trials are not designed to prove efficacy, the ABACUS-1 study included several exploratory assessments to notice if KIO-301 was having a functional effect. These included visual acuity tests, kinetic visual fields, and functional magnetic resonance imaging (fMRI).

One of the most compelling findings came from the fMRI data. Researchers observed light-evoked blood-oxygen-level-dependent (BOLD) signal changes in the visual cortical regions of the brain following the dosing of KIO-301. These signal changes showed a temporal pattern compatible with pharmacodynamic activity, suggesting that the “photoswitch” was successfully triggering a response in the brain’s visual processing centers according to the Nature Medicine publication.
some participants showed variation in light perception and functional vision measures. However, these results were varied across the small group, highlighting the need for a more controlled environment to determine if these changes translate into a reliable, everyday benefit for the patient.
The Path to Phase 2: ABACUS-2
The successful safety profile of the ABACUS-1 trial has paved the way for the next stage of clinical development. Kiora Pharmaceuticals (NASDAQ: KPRX) has already initiated a randomized, controlled Phase 2 clinical trial, referred to as ABACUS-2 as announced by the company.
The transition to a randomized, controlled trial is a critical step. It will allow researchers to compare the effects of KIO-301 against a control group, providing a clearer picture of whether the light responsiveness observed in the exploratory phase of ABACUS-1 is a direct result of the therapy. This phase will be essential in determining if the treatment can provide a meaningful improvement in the quality of life for those with advanced retinal degeneration.
Dr. Robert Casson, the Principal Investigator from the Royal Adelaide Hospital, emphasized the importance of this progression. He noted that while the Phase 1 findings provide “evidence of short-term ocular safety and feasibility,” larger and more controlled studies are necessary to confirm if the functional changes provide a “reliable, everyday vision benefit” per the trial’s publication details.
Comparison of Trial Phases
| Feature | ABACUS-1 (Phase 1) | ABACUS-2 (Phase 2) |
|---|---|---|
| Primary Goal | Safety and Feasibility | Efficacy and Functional Benefit |
| Study Design | Open-label, Dose-escalation | Randomized, Controlled |
| Participants | 6 participants (12 eyes) | Larger participant cohort |
| Key Finding | Acceptable safety profile; BOLD signal changes | TBD (Underway) |
What This Means for Patients
For the global community of people with retinitis pigmentosa, the gene-agnostic nature of KIO-301 is particularly hopeful. Many inherited retinal diseases are caused by a vast array of different genetic mutations, making it tricky to develop a single “cure” that works for everyone. A therapy that targets the retinal ganglion cells rather than the specific mutation offers a broader potential application.

However, it is key to manage expectations. The ABACUS-1 trial was a small, nonrandomized study. While the safety results are encouraging and the fMRI signals are a positive indicator of brain activity, the therapy is still in the early stages of clinical testing. The goal is not necessarily a full restoration of 20/20 vision, but rather the restoration of enough light perception to improve mobility, orientation, and independence.
As we move forward, the medical community will be watching the ABACUS-2 trial closely to see if the exploratory signals of light responsiveness can be replicated and quantified in a larger population. This research represents a bold attempt to redefine the boundaries of treatable blindness.
The next confirmed checkpoint for this therapy is the progression and subsequent data release from the ongoing ABACUS-2 randomized, controlled Phase 2 clinical trial.
Do you or a loved one live with retinitis pigmentosa? We welcome your thoughts and experiences in the comments below, and encourage you to share this update with others in the patient community.