The 340B Drug Pricing Program, designed to help safety-net providers stretch scarce federal resources to serve vulnerable patients, is facing renewed scrutiny as evidence mounts that its discounts are increasingly benefiting institutions rather than the patients it was meant to serve. The American College of Physicians (ACP) has entered the debate with a new policy brief calling for urgent reforms to restore the program’s original intent and safeguard its future.
In a statement released alongside the publication of their policy brief in Annals of Internal Medicine, the ACP emphasized that without meaningful changes, the 340B program risks eroding public trust and failing to deliver on its promise to improve access to affordable medications for low-income and uninsured populations. The organization’s recommendations focus on increasing transparency, strengthening oversight, and redirecting savings directly to patient care.
The policy brief, titled “Reforming 340B to Promote Program Integrity and Better Serve Vulnerable Populations,” outlines a series of evidence-based proposals aimed at curbing profit-seeking behaviors by hospitals and clinics that participate in the program. According to the ACP, current loopholes allow some providers to purchase drugs at deeply discounted 340B prices while charging patients and insurers near-market rates, pocketing the difference as revenue.
This practice, often referred to as “spread pricing,” has drawn criticism from patient advocacy groups and policymakers who argue it undermines the program’s mission. The ACP contends that such diverging incentives must be realigned through structural reforms that prioritize patient outcomes over financial gain.
Understanding the 340B Program and Its Original Purpose
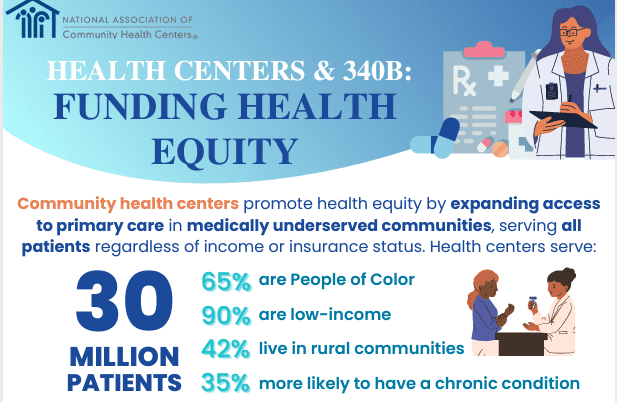
Established in 1992 under Section 340B of the Public Health Service Act, the 340B Drug Pricing Program requires pharmaceutical manufacturers participating in Medicaid to offer outpatient drugs to eligible healthcare organizations at significantly reduced prices. The program was created to help federal grantees — such as community health centers, disproportionate share hospitals, and other safety-net providers — stretch limited federal resources to reach more patients in need.
Eligible institutions can purchase covered outpatient drugs at prices that are typically 20% to 50% below the average manufacturer price, depending on the medication. These savings are intended to be reinvested into patient services, including free or low-cost medication distribution, outreach programs, and supportive care for uninsured or underinsured individuals.
Over the past decade, however, the program has expanded significantly, with more than 34,000 contract pharmacy arrangements now in place across the United States. This growth has coincided with rising concerns about whether the financial benefits are reaching patients or being absorbed by hospital systems as unrestricted revenue.
ACP’s Evidence-Based Recommendations for Reform
The ACP’s policy brief does not call for dismantling the 340B program but instead advocates for targeted reforms to strengthen accountability and ensure that discounts translate into tangible benefits for patients. Key recommendations include:
- Requiring greater transparency in how 340B savings are used, including standardized reporting on patient assistance programs and charity care.
- Implementing stricter oversight mechanisms to prevent duplicate discounts and diversion of medications meant for eligible patients.
- Revising eligibility criteria to focus funding on providers that serve a high proportion of low-income and uninsured patients.
- Encouraging the leverage of 340B savings to support innovative care models, such as medication therapy management and chronic disease prevention programs.
The organization stresses that any reform effort must balance program integrity with continued access for vulnerable populations, warning that overly restrictive changes could inadvertently harm the very communities the program seeks to help.
Stakeholder Perspectives and Ongoing Debates
The ACP’s position adds to a growing chorus of voices calling for 340B reform. In recent months, both the Department of Health and Human Services (HHS) and the Government Accountability Office (GAO) have released reports highlighting weaknesses in program oversight and recommending greater federal scrutiny.
Meanwhile, hospital associations and some pharmaceutical companies have defended the current structure, arguing that 340B revenues are essential for maintaining services in underserved areas, particularly rural hospitals facing financial strain. They caution that reducing program benefits could lead to cutbacks in essential services rather than improved patient outcomes.
Patient advocacy groups, however, have largely supported the ACP’s stance, citing instances where patients eligible for 340B discounts still face high out-of-pocket costs due to opaque billing practices. These groups advocate for clearer guidelines that would require providers to pass on savings directly at the point of sale.
What Comes Next: Policy Pathways and Implementation
As of April 2022, the ACP’s policy brief represents one of the most comprehensive clinical organization-led analyses of the 340B program to date. While no federal legislation has yet been enacted based on these recommendations, the brief has been submitted to congressional committees overseeing healthcare policy, including the House Energy and Commerce Committee and the Senate Finance Committee.
The next steps in the reform process will likely involve further stakeholder consultations, potential rulemaking by the Health Resources and Services Administration (HRSA), which administers the program, and possible legislative action during upcoming congressional sessions. The ACP has indicated it will continue to engage with policymakers to advocate for evidence-based changes that uphold the program’s foundational goals.
For individuals seeking updated information on the 340B program, official resources are available through the HRSA website, including program guidelines, eligibility criteria, and annual reports. The ACP also encourages clinicians and patients to stay informed through professional medical societies and public health advocacy organizations.
As discussions around healthcare affordability and equity continue to evolve, the future of the 340B program remains a critical test of whether well-intentioned policies can be adapted to serve their original purpose in a changing healthcare landscape.
We invite our readers to share their perspectives on this important issue. Have you or someone you know interacted with the 340B program as a patient or provider? What changes would you like to see to ensure it better serves vulnerable populations? Join the conversation in the comments below and help spread awareness by sharing this article with others interested in healthcare policy and patient advocacy.