For years, the disparity between what Americans pay for prescription medications and the prices found in other developed nations has been a point of contention in public health. For the millions of individuals relying on Medicaid, these costs are not just line items in a federal budget; they represent the difference between consistent treatment and precarious gaps in care.
In a strategic move to address these systemic imbalances, the Centers for Medicare & Medicaid Services (CMS) has introduced the GENEROUS Model. Designed to lower GENEROUS Model Medicaid drug costs, this initiative seeks to ensure that the prices paid for essential medications are both fair and reasonable through a new framework of CMS-led negotiations with drug manufacturers.
As a physician and health journalist, I have watched the sustainability of public health programs struggle under the weight of escalating pharmacy expenditures. The GENEROUS (GENErating cost Reductions fOr U.S. Medicaid) Model represents a shift toward international price alignment, aiming to protect the most vulnerable populations while easing the financial burden on state budgets.
How the GENEROUS Model Works
The core mechanism of the GENEROUS Model is the implementation of supplemental rebates. Under this voluntary program, participating drug manufacturers will provide these rebates to states to align Medicaid net prices with the costs paid in other select countries. By utilizing international drug prices as a benchmark, CMS intends to curb the inflation of prescription drug spending within the Medicaid program.
Unlike traditional pricing structures, this model relies on CMS-led negotiations to determine the level of supplemental rebates. This approach allows the federal government to leverage its collective bargaining power to secure prices that more closely mirror the global market, rather than leaving price discovery solely to individual state negotiations or standard rebate formulas.
The model is designed to be flexible and voluntary for both the manufacturers of the drugs and the states that administer the Medicaid programs. This ensures that participation is driven by mutual agreement and a shared goal of increasing healthcare sustainability.
The Financial Urgency Behind the Initiative
The drive toward the GENEROUS Model is fueled by a stark increase in pharmacy spending. According to data from the CMS Innovation Center, net spending for Medicaid drugs reached $60 billion in 2024. This represents a significant increase of $10 billion from the spending levels recorded in 2022.

This trajectory is unsustainable for many state governments. When a disproportionate share of a health budget is consumed by rising drug costs, there is often less flexibility to invest in other critical resources, such as preventative care, mental health services, or primary care infrastructure. By reducing the net price of medications, the GENEROUS Model aims to provide states with the fiscal breathing room necessary to improve overall health outcomes.
Beyond the balance sheets, the model is fundamentally about patient access to medications. When drug prices are lowered, the potential for broader coverage of needed medications increases, ensuring that patients with chronic conditions or acute needs do not face barriers to the therapies they require.
Timeline for Implementation and Participation
The rollout of the GENEROUS Model follows a strict timeline to ensure that both manufacturers and states have adequate time to evaluate the Request for Applications (RFA). The model is scheduled to officially launch in January 2026 and will operate for a period of five years.
For stakeholders looking to participate, CMS has established specific deadlines for application. The deadline for drug manufacturers to apply to the model has been extended to June 11, 2026. States interested in participating in the model have until September 10, 2026, to submit their State Request for Applications.
| Milestone | Deadline/Date |
|---|---|
| Model Official Launch | January 2026 |
| Manufacturer Application Deadline | June 11, 2026 |
| State Application Deadline | September 10, 2026 |
| Program Duration | Five Years |
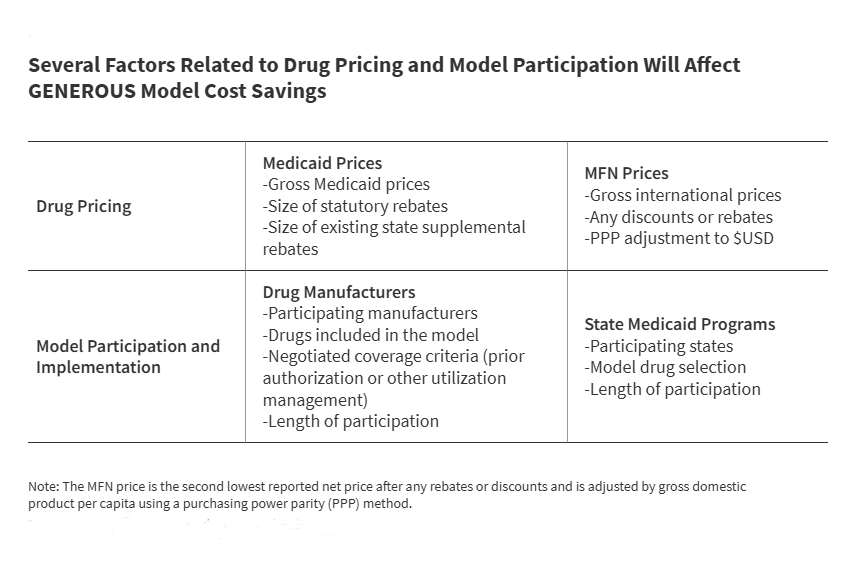
Factors Influencing the Model’s Overall Impact
While the goals of the GENEROUS Model are clear, its ultimate success will depend on several variables, some of which remain confidential or uncertain. Because the model is voluntary, the total savings will be directly tied to the number of manufacturers and the specific drugs that are included in the program.
Key factors that will determine the impact on Medicaid drug costs include:
- Manufacturer Adoption: The extent to which pharmaceutical companies agree to the supplemental rebate structure.
- State Enrollment: The number of states that opt into the model to leverage the negotiated prices.
- Drug Selection: Whether the model covers high-cost specialty drugs, which often drive the bulk of spending increases, or focuses on a broader range of generic and brand-name medications.
- International Price Volatility: Since the model aligns prices with other countries, fluctuations in international pricing could influence the value of the supplemental rebates.
The CMS Innovation Center is tasked with balancing these factors to ensure that the pursuit of lower costs does not inadvertently lead to drug shortages or reduced manufacturer investment in the U.S. Market. The focus remains on achieving “fair and reasonable” prices that support both the patient and the provider.
Key Takeaways
- Objective: To reduce Medicaid drug spending by aligning U.S. Net prices with international benchmarks.
- Mechanism: CMS-led negotiations leading to supplemental rebates from manufacturers to participating states.
- Financial Context: Medicaid drug spending rose to $60 billion in 2024, a $10 billion increase over two years.
- Participation: The model is voluntary for both states and drug manufacturers.
- Duration: A five-year initiative launching in January 2026.
As we move toward the 2026 launch, the healthcare community will be watching closely to see which manufacturers step forward and which states prioritize this path to sustainability. The ability to decouple essential medicine from prohibitive pricing is a critical step in modernizing public health infrastructure.
The next major checkpoint for the program is the manufacturer application deadline on June 11, 2026. We will continue to monitor the enrollment numbers and the list of included medications as they become available.
Do you believe international price benchmarking is the right approach for the U.S. Healthcare system? Share your thoughts in the comments below or share this article with your professional network.