As the scientific community grapples with evolving models for preclinical research, a growing emphasis is being placed on integrating diverse experimental systems rather than choosing between them. The conversation around translational science is shifting from debates about animal versus human or AI models toward strategies that combine approaches to strengthen confidence in findings, enhance relevance to human biology, and improve decision-making in drug development and disease research.
This integrated perspective reflects broader trends in biomedical innovation, where researchers are leveraging complementary strengths across models to address complex questions. Animal models continue to play a role in studying systemic physiology and long-term outcomes, while human-derived systems such as organoids offer insights into human-specific biology and pathology. Meanwhile, artificial intelligence and computational models are increasingly used to predict responses, analyze large datasets, and reduce reliance on empirical testing alone.
The goal, as articulated in recent discussions within the field, is not to replace one system with another but to thoughtfully combine them so that each contributes to a more robust understanding of biological mechanisms and potential interventions. This approach aims to increase the predictive value of preclinical work while addressing ethical and practical considerations associated with any single model system.
Recent advances illustrate how this integration can work in practice. For example, studies involving human airway organoids have demonstrated their utility in studying highly pathogenic viruses under biosafety level 4 conditions, providing a human-relevant platform that complements animal studies and informs public health responses. These models allow scientists to examine viral replication and host responses in human tissue without the require for live infectious agents in human subjects.
Similarly, research into therapeutic development for complex conditions like snakebite envenoming has benefited from the use of large animal models, which better mimic human physiology and pharmacokinetics than smaller species. Such models are critical for evaluating the safety and efficacy of potential antidotes before human trials, offering a bridge between mechanistic studies and clinical application.
In maternal-fetal medicine, bioengineering approaches are being combined with animal and human data to innovate solutions for pregnancy-related complications. Engineers and clinicians are collaborating to design devices, biomaterials, and diagnostic tools that are tested across multiple systems to ensure both functionality and biological compatibility.
These examples underscore a growing consensus that the most reliable preclinical insights emerge not from isolating one model type but from strategically aligning animal, human-based, and computational systems to answer specific scientific questions. Each model has inherent limitations—animal models may not fully recapitulate human disease, human organoids lack systemic circulation and immune components, and AI models depend on the quality and breadth of training data—but together, they can compensate for individual weaknesses.
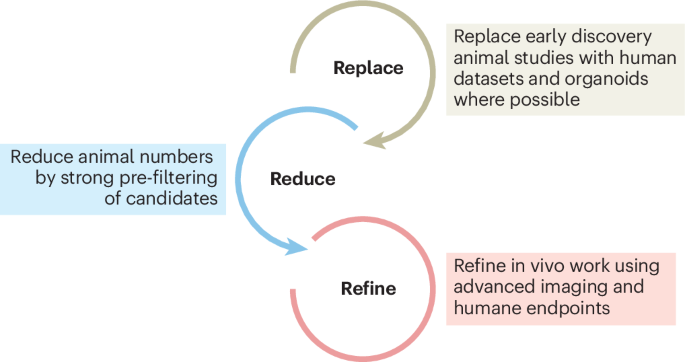
This systems-based approach also supports the principles of the 3Rs (replacement, reduction, refinement) in animal research by enabling scientists to use animal models more strategically, often in later stages of investigation after preliminary screening in human-derived or computational systems. It encourages a more nuanced evaluation of when and how animal studies add unique value that cannot be obtained otherwise.
Funding agencies and research institutions are beginning to reflect this shift in priorities, supporting projects that explicitly plan for cross-model validation and mechanistic triangulation. There is increasing recognition that granting frameworks and publication standards should encourage, rather than discourage, integrated methodologies that enhance rigor and translatability.
Moving forward, the success of such integrated preclinical packages will depend on clear communication between disciplines, standardized methods for comparing results across systems, and shared access to data and models. Collaborative platforms that bring together experts in biology, engineering, computational science, and clinical medicine are seen as essential to driving this evolution in research practice.
As the field continues to evolve, the focus remains on building preclinical evidence that is not only scientifically sound but also genuinely useful for informing real-world decisions about health interventions. By combining the strengths of multiple approaches, researchers aim to produce knowledge that is more reliable, more human-relevant, and better equipped to support the development of safe and effective therapies.
For ongoing updates on advances in preclinical research models and translational science, readers can follow developments through major scientific publishers and biomedical research institutions that regularly report on emerging methodologies and validation studies.