Targeted Therapies Plus Radiotherapy for Diffuse Intrinsic Pontine Glioma: Insights from the BIOMEDE Trial
In a significant development for pediatric oncology, results from the biomarker-driven phase 2 BIOMEDE trial have clarified the limited benefit of adding targeted therapies to standard radiotherapy for diffuse intrinsic pontine glioma (DIPG), while identifying biological features associated with longer survival. Published in Nature Medicine on April 24, 2026, the study evaluated radiotherapy combined with erlotinib, everolimus, or dasatinib in children newly diagnosed with DIPG, a highly aggressive brainstem tumor with a poor prognosis. Although the primary endpoint of overall survival was not met across any treatment arm, researchers identified molecular and histological characteristics linked to improved outcomes, with everolimus showing suggestive activity warranting further investigation.
DIPG, also known as diffuse midline glioma, primarily affects children between the ages of 5 and 10 and accounts for approximately 10-15% of all childhood brain tumors. Due to its location in the pons, surgical resection is not feasible and radiotherapy remains the cornerstone of treatment, typically providing only transient symptom relief. Median survival following diagnosis is less than one year, and fewer than 10% of patients survive two years. The lack of therapeutic progress over decades has driven interest in molecularly guided approaches, particularly given the tumor’s frequent histone H3 mutations and activation of signaling pathways such as PI3K/mTOR and EGFR.
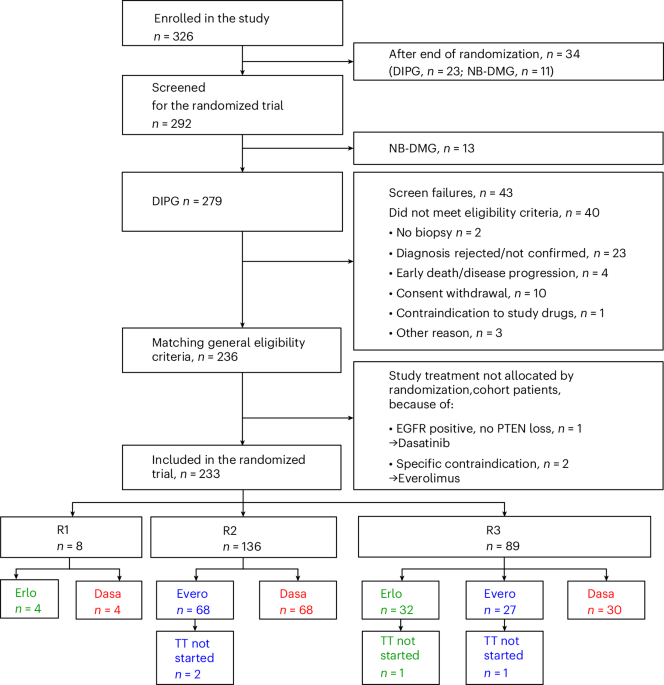
The BIOMEDE trial (Biological Medicine for DIPG Eradication), conducted across multiple centers in France and supported by international collaborators, enrolled 89 patients with newly diagnosed, radiologically confirmed DIPG between 2014 and 2018. Participants were stratified based on tumor biopsy results and assigned to receive standard radiotherapy (54 Gy in 30 fractions) plus one of three targeted agents: erlotinib (an EGFR inhibitor), everolimus (an mTOR inhibitor), or dasatinib (a dual SRC/ABL inhibitor). The primary objective was overall survival at two years, with secondary endpoints including progression-free survival, biomarker correlations, and safety.
According to the published results, no significant improvement in overall survival was observed in any treatment group compared to historical controls receiving radiotherapy alone. The two-year survival rate remained below 10% across all arms, underscoring the persistent therapeutic challenge posed by DIPG. However, exploratory analyses revealed that certain tumor characteristics were associated with longer survival. Patients whose tumors exhibited histone H3 wild-type status or lacked the H3K27M mutation demonstrated improved outcomes, as did those with lower tumor cell density on histopathological examination. Radiographic evidence of contrast enhancement—often linked to blood-brain barrier disruption—was correlated with survival beyond 18 months.
Among the targeted agents, everolimus showed the most promising signal, with a subset of patients experiencing prolonged stable disease or modest tumor regression. While not sufficient to meet the primary endpoint, this observation prompted researchers to suggest that mTOR pathway inhibition may warrant further evaluation in biomarker-selected populations, particularly those with PI3K/mTOR pathway activation identified through genomic profiling. Erlotinib and dasatinib did not demonstrate comparable activity in this cohort.
The trial also contributed to evolving classification systems for diffuse gliomas by reinforcing the importance of molecular diagnostics. As noted in recent research, copy-number alterations and histone mutation status are increasingly recognized as critical factors in refining diagnosis and predicting behavior in pediatric high-grade gliomas. These findings support the shift toward biologically defined categories rather than relying solely on histology or location, a transition formalized in the 2021 update to the World Health Organization classification of central nervous system tumors.
Experts emphasize that while the BIOMEDE trial did not yield a new standard of care, it provided valuable insights into tumor biology and the limitations of broad molecular targeting in heterogeneous diseases like DIPG. The results reinforce the need for precision approaches that match therapies to specific molecular alterations, rather than applying agents based on pathway assumptions alone. Ongoing efforts are focusing on combination strategies, immune-based therapies, and novel delivery methods such as convection-enhanced delivery or nanoparticle-mediated transport to overcome the blood-brain barrier.
Families affected by DIPG continue to advocate for increased research funding and access to clinical trials through organizations such as The Cure Starts Now and the DIPG Registry, which collect clinical and molecular data to accelerate discovery. Ongoing trials are exploring agents targeting epigenetic regulators, GD2-directed immunotherapy, and oncolytic viruses, with early-phase results showing cautious optimism in select subgroups.
The next major update in DIPG research is expected from the International Symposium on Pediatric Neuro-Oncology, scheduled for September 2026 in Heidelberg, Germany, where long-term follow-up from trials like BIOMEDE and emerging therapeutic strategies will be presented. For now, radiotherapy remains the foundation of care, with treatment decisions guided by multidisciplinary evaluation and, increasingly, molecular profiling where available.
We invite our readers to share thoughts, experiences, or questions in the comments below. If you found this article informative, please consider sharing it to help raise awareness about pediatric brain tumor research.