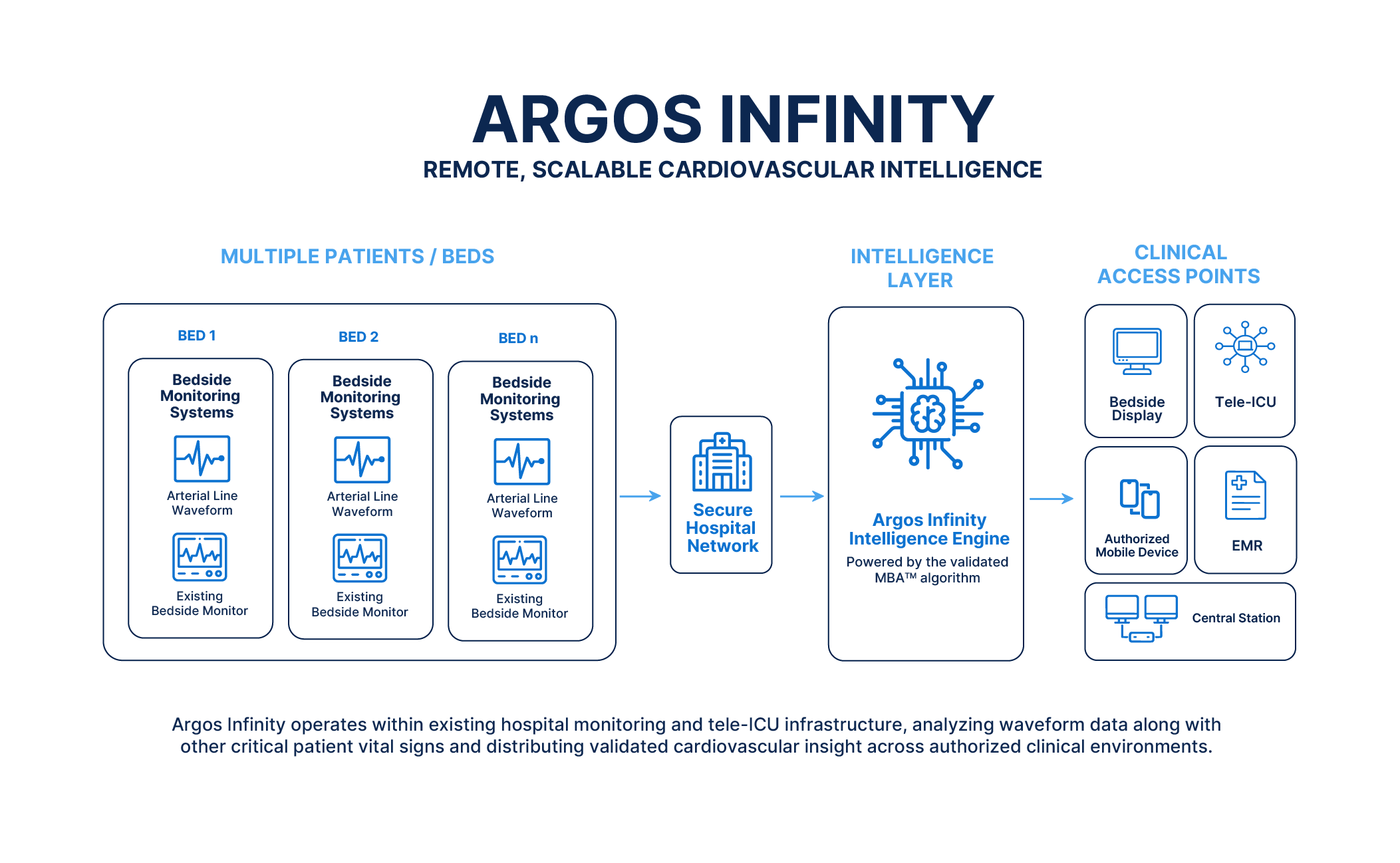
In critical care settings, timely detection of cardiovascular deterioration can mean the difference between life and death. Yet, traditional hemodynamic monitoring often remains confined to bedside hardware, limiting access to real-time insights when and where clinicians need them most. This gap in visibility has driven innovation in medical technology, with companies like Retia Medical pioneering software-driven solutions designed to transform how cardiovascular data is collected, analyzed, and acted upon across hospital environments.
At the forefront of this shift is Marc Zemel, CEO of Retia Medical, whose personal experience with sudden cardiac loss fueled a mission to improve cardiovascular monitoring through distributed intelligence. Rather than relying solely on invasive or isolated hardware systems, Retia’s approach leverages existing hospital data streams to deliver advanced hemodynamic insights via software platforms like Argos Infinity. This model aims to expand access to critical cardiovascular information not just in operating rooms and intensive care units, but similarly in tele-ICU settings and remote monitoring scenarios.
Recent regulatory milestones underscore the progress of this vision. In April 2026, Marc Zemel shared on LinkedIn that he had recently spoken with Saul W. Marquez on the Outcomes Rocket podcast about Retia Medical’s FDA clearance of Argos Infinity, describing it as a cardiovascular intelligence software platform designed to help clinicians detect deterioration earlier and improve outcomes in high-risk surgical and critical care environments.
The Argos Infinity platform received FDA 510(k) clearance, a fact confirmed through Retia Medical’s official press release and documented in the FDA’s premarket notification database under submission number K181372. The device, classified as a Computer, Diagnostic, Pre-Programmed, Single-Function system under product code DXG, was reviewed by the Cardiovascular panel and determined to be substantially equivalent to legally marketed predicate devices. The decision date for this clearance was December 13, 2018, establishing the regulatory foundation for subsequent iterations and clinical deployment.
Retia Medical, headquartered in Valhalla, New York, continues to advance its mission of redefining cardiovascular monitoring by moving beyond hardware-centric models. As Zemel explained in a featured appearance on The State of Medtech podcast, the company’s focus is on creating distributed intelligence that enables hospitals to do more with fewer resources—particularly valuable in settings facing staffing constraints or geographic limitations. By analyzing data already collected within clinical workflows, Argos Infinity aims to provide actionable insights without requiring additional invasive monitoring.
This approach addresses a persistent challenge in critical care: the delayed recognition of low cardiac index despite normal blood pressure readings. A study published in Anesthesia & Analgesia and referenced in Retia Medical’s press materials demonstrated the frequent occurrence of undetected hemodynamic instability using the Argos® Monitor, highlighting how traditional vital signs can mask underlying cardiovascular compromise. Earlier detection through continuous, software-driven monitoring could reduce delays in intervention, potentially lowering morbidity and mortality associated with postoperative or septic cardiovascular deterioration.
The clinical implications extend beyond individual patient outcomes. Hospitals adopting distributed cardiovascular intelligence may notice improved efficiency in care delivery, reduced reliance on specialized monitoring equipment, and enhanced capacity for remote consultations through tele-ICU integration. These benefits align with broader healthcare trends toward scalable, data-driven solutions that support clinical decision-making without increasing procedural burden.
Looking ahead, Retia Medical’s strategy includes expanding access to Argos Infinity through strategic partnerships, such as its US distribution agreement with Medtronic for the Argos® Cardiac Output Monitor. Such collaborations aim to broaden reach across diverse care settings whereas maintaining regulatory compliance and clinical validity. Ongoing studies and real-world evaluations will be critical in assessing long-term impact on patient safety, resource utilization, and clinical workflow integration.
For healthcare professionals seeking updates on Retia Medical’s developments, including clinical evidence, regulatory status, and product information, the company maintains an active presence on LinkedIn and provides press releases and technical details through its official website. The FDA’s 510(k) database remains a primary source for verifying the clearance status of Argos Infinity and related devices under submission K181372.
As the medical technology landscape evolves toward software-defined, interoperable systems, innovations like distributed cardiovascular intelligence represent a meaningful step toward more proactive, accessible, and resilient critical care. By transforming existing data into timely clinical insights, platforms such as Argos Infinity aim to ensure that the right information reaches the right clinician at the right time—before deterioration becomes irreversible.
Stay informed about advances in medical technology and patient monitoring by following trusted regulatory updates and peer-reviewed clinical research. Share your perspectives on how software-driven innovations are shaping the future of critical care in the comments below.